Antoine Lavoisier, John Dalton, Jöns Jacob Berzelius, Dmitri Mendeleev, and Marie Curie
Let’s provide concise summaries of the key contributions and significance of Antoine Lavoisier, John Dalton, Jöns Jacob Berzelius, Dmitri Mendeleev, and Marie Curie, focusing on their impact on the development of chemistry and highlighting their distinct achievements.
- Antoine Lavoisier (1743-1794) – The Quantitative Revolution:
- Nationality: French
- Key Contributions:
- Combustion and Oxygen: He definitively disproved the phlogiston theory, demonstrating that combustion involves the combination of a substance with oxygen (which he named). This was a fundamental shift in understanding chemical reactions.
- Law of Conservation of Mass: He rigorously established that mass is conserved in chemical reactions – a cornerstone of quantitative chemistry. He showed that matter might change form, but its total mass remains unchanged.
- Chemical Nomenclature: He developed a systematic nomenclature for chemical compounds (e.g., “sulfuric acid” instead of “oil of vitriol”), replacing the older, inconsistent system. This standardized chemical communication.
- Elements vs. Compounds: He clearly distinguished between elements and compounds and created an early list of elements.
- Elements of Chemistry (Textbook): His textbook systematized his discoveries and presented the new chemistry clearly and organized, influencing generations of chemists.
- Significance: Lavoisier is considered the “father of modern chemistry” because he transformed it from a qualitative, descriptive science (often bordering on alchemy) into a quantitative, experimental science based on precise measurement and the conservation of mass. He established the foundation for modern chemical theory, and his emphasis on careful experimentation and measurement revolutionized the field.
- John Dalton (1766-1844) – The Atomic Hypothesis:
- Nationality: English
- Key Contributions:
- Atomic Theory (1803-1808): Dalton’s atomic theory is his defining achievement. He proposed that:
- All matter is made of atoms, which are indivisible and indestructible. (We now know atoms are divisible, but this was a crucial step.)
- All atoms of a given element are identical in mass and properties. (We now know about isotopes).
- Compounds are formed by combining two or more different kinds of atoms.
- A chemical reaction is a rearrangement of atoms.
- Law of Multiple Proportions: He observed that when elements combine to form different compounds, they do so in ratios of small whole numbers (e.g., carbon and oxygen can form CO or CO2, but not CO1.5). This provided strong evidence for his atomic theory.
- First Table of Atomic Weights: He created the first table of relative atomic weights, assigning weights to each known element (though many of his values were inaccurate).
- Gas Laws: He also studied the behavior of gases and developed Dalton’s Law of Partial Pressures.
- Atomic Theory (1803-1808): Dalton’s atomic theory is his defining achievement. He proposed that:
- Significance: Dalton’s atomic theory provided the conceptual framework for modern chemistry. It explained chemical reactions as the rearrangement of atoms and provided a basis for understanding the composition of matter. While some details of his theory were later modified, the core idea of atoms as the fundamental building blocks of matter remains central to chemistry.
- Jöns Jacob Berzelius (1779-1848) – Systematization and Notation:
- Nationality: Swedish
- Key Contributions:
- Modern Chemical Notation: He introduced the system of using letters to represent elements (e.g., O for oxygen, Fe for iron, etc.), and he developed the basic form of chemical formulas we use today (e.g., H2O for water).
- Accurate Atomic Weights: He painstakingly determined the atomic weights of many elements with much greater accuracy than Dalton, providing essential data for chemical calculations.
- Discovery of Elements: He discovered several new elements: cerium, selenium, and thorium.
- Electrochemical Dualism: He proposed an early electrochemical theory of chemical bonding, suggesting that the attraction of oppositely charged atoms formed compounds.
- Isomerism and Catalysis: He coined the terms “isomerism” (compounds with the same formula but different structures) and “catalysis” (substances that speed up reactions without being consumed).
- Significance: Berzelius brought systematic organization to chemistry. His chemical notation made it much easier to represent and understand chemical reactions. His accurate atomic weights were crucial for quantitative analysis and the development of chemical theory. He was a dominant figure in chemistry in the first half of the 19th century, and his textbooks and influence shaped the field.
- Dmitri Mendeleev (1834-1907) – The Periodic System:
- Nationality: Russian
- Key Contributions:
- Periodic Table (1869): Mendeleev arranged the known elements in order of increasing atomic weight (later refined to atomic number) and observed that elements with similar chemical properties appeared at regular intervals. He created the periodic table, leaving gaps for undiscovered elements.
- Periodic Law: He formulated the periodic law, stating that the properties of elements are periodic functions of their atomic weights.
- Predictions: Based on the gaps in his table, he predicted the existence and properties of several undiscovered elements (gallium, scandium, germanium). The later discovery of these elements, with properties remarkably close to his predictions, was a stunning validation of his work.
- Significance: The periodic table is one of chemistry’s most important organizing principles. It provides a framework for understanding the relationships between elements, predicting their properties, and understanding chemical reactions. It’s a fundamental tool for all chemists and a testament to the power of scientific prediction.
- Marie Curie (1867-1934) – Radioactivity:
- Nationality: Polish-French
- Key Contributions:
- Discovery of Radioactivity: Along with her husband, Pierre Curie, she investigated the phenomenon of radioactivity (a term she coined), discovering that certain elements (uranium, thorium) spontaneously emit radiation.
- Discovery of Polonium and Radium (1898): She isolated two new radioactive elements, polonium (named after her native Poland) and radium, from pitchblende ore. This was a painstaking and arduous process.
- Development of Techniques for Isolating Radioactive Isotopes: She developed methods for separating and purifying radioactive materials.
- Nobel Prizes: Awarded the Nobel Prize in Physics in 1903 (shared with Pierre Curie and Henri Becquerel) for their work on radioactivity and the Nobel Prize in Chemistry in 1911 for discovering polonium and radium.
- Significance: Curie’s work on radioactivity revolutionized physics and chemistry. It challenged the idea of the atom as indivisible and unchanging, opening up the field of nuclear physics. Her discoveries had profound implications for medicine (radiotherapy), industry, and our understanding of the fundamental nature of matter. She was a pioneering scientist who overcame significant gender barriers to achieve groundbreaking discoveries.
These five chemists, through their theoretical insights, experimental discoveries, and organizational frameworks, fundamentally transformed the field of chemistry and laid the groundwork for modern science. They represent different eras and different approaches, but they all share a commitment to rigorous inquiry and a profound impact on our understanding of the world.
Antoine (1743-1794) and his Wife Marie-Anne (1758–1836) Lavoisier – The Quantitative Revolution

Portrait of Lavoisier explaining to his wife Marie-Anne the result of his experiments on air by Ernest Board. She was to play an essential part in Lavoisier’s scientific career—notably, she translated English documents for him, including Richard Kirwan‘s Essay on Phlogiston and Joseph Priestley‘s research. In addition, she assisted him in the laboratory and created many sketches and carved engravings of the laboratory instruments used by Lavoisier and his colleagues for their scientific works.
(Wiki Image By https://wellcomeimages.org/indexplus/obf_images/1b/c2/02875d4a50a175cf155b7b6230b0.jpgGallery: https://wellcomecollection.org/works/urc4max8 CC-BY-4.0, CC BY 4.0, https://commons.wikimedia.org/w/index.php?curid=36515225)
Lavoisier Quotes Table
Finding precisely documented, verbatim quotes from Antoine Lavoisier in English translation can be tricky. Many quotes are paraphrased or taken from his writings, originally in French. However, I can provide a table with some of his most significant and representative statements, along with context and explanation, clearly indicating the source and nature of each quote.
| Quote Number | Quote | Source | Context/Explanation | Significance |
| 1 | “Nothing is lost, nothing is created, all is transformed.” ( Rien ne se perd, rien ne se crée, tout se transforme.) | Often paraphrased from Traité Élémentaire de Chimie (1789) | It expresses the principle of conservation of mass in chemical reactions, a cornerstone of Lavoisier’s work and modern chemistry. He didn’t state it in this exact form, but the idea is central to his work. | Fundamental principle of chemistry. Demonstrates that matter is not created or destroyed in chemical reactions but only changes form. |
| 2 | “…it is impossible to separate the nomenclature of a science from the science itself…” | Méthode de Nomenclature Chimique (1787) | From his work on chemical nomenclature, written with other chemists. Highlights the importance of clear and consistent language for scientific progress. | Lavoisier helped develop a systematic way to name chemical compounds, crucial for clear communication and understanding of chemistry. |
| 3 | “I have tried…to arrive at the truth by linking up facts, by suppressing as much as possible the use of reasoning, which is always a treacherous instrument.” | Traité Élémentaire de Chimie (1789) (Preface – paraphrased) | This reflects his emphasis on empirical observation and experimentation as the basis for scientific knowledge and his distrust of purely theoretical speculation. | This highlights his commitment to the scientific method and his focus on experimental evidence, which was a major departure from the more philosophical and speculative approaches of earlier chemistry (and alchemy). |
| 4 | “[On air]…it became evident to me that a portion of the air…combines with metals during calcination…” | (Paraphrased from his memoirs on combustion) | Describes his key experimental finding that a component of air (oxygen) combines with metals when they are heated (calcined), leading to an increase in weight. | This observation directly contradicted the phlogiston theory and was crucial for his development of the oxygen theory of combustion. |
| 5 | “[On respiration]…Respiration is therefore a combustion, slow it is true, but otherwise perfectly similar to that of charcoal.” | (Paraphrased from his work on respiration) | Shows his insight that respiration is a process similar to combustion, involving oxygen consumption and energy release. | It connects chemistry to biology, demonstrating the fundamental chemical processes that underlie life. |
| 6 | “The art of drawing conclusions from experiments and observations consists in evaluating probabilities…” | Traité élémentaire de chimie | Shows understanding of evidence. |
Export to Sheets
Key:
- Paraphrased: Indicates that the quote is a close paraphrase of Lavoisier’s words, based on his writings, but may not be a perfectly verbatim translation.
- Representative: Indicates a statement that accurately reflects his known views and scientific approach, even if not a direct quote.
- Traité Élémentaire de Chimie: Lavoisier’s Elements of Chemistry, a highly influential textbook that presented his new theories and nomenclature.
Key Takeaways:
- Emphasis on Experimentation: Lavoisier’s quotes highlight his commitment to careful observation, precise measurement, and drawing conclusions based on experimental evidence.
- Quantitative Approach: He stressed the importance of quantifying chemical phenomena, a major shift from earlier, more qualitative approaches.
- Conservation of Mass: The principle of conservation of mass is central to his work and is reflected in his writings.
- Clarity of Language: He recognized the importance of clear and consistent language for scientific progress, leading him to develop a new chemical nomenclature system.
- Foundations of Modern Chemistry: These quotes, taken together, illustrate how Lavoisier’s work laid the foundations for modern chemistry.
This table provides a structured and informative overview of key statements by and about Antoine Lavoisier. It highlights his emphasis on experimentation, quantification, and clear communication, all crucial to his revolutionary contributions to chemistry. The table clarifies the sourcing and nature of each quote, providing context and explanation for its significance.
Lavoisier YouTube Video
- Antoine Lavoisier and the Origin of Modern Chemistry | AMS OpenMind by OpenMind: 234,235 views (http://www.youtube.com/watch?v=55yJPs-_R1w)
- How the French Revolution Nearly Destroyed Modern Chemistry by Weird History: 174,604 views (http://www.youtube.com/watch?v=zFzu2y0DOZ8)
- Antoine Lavoisier Biography by CloudBio: 51,720 views (http://www.youtube.com/watch?v=ZgmGxN66rTQ)
- Final Experiment: The Execution of Lavoisier #Lavoisier #HistoryOfScience #HistoricalExperiments by Spheres of Secrets: 10,391 views (http://www.youtube.com/watch?v=z2i-tN1N7DI)
- Antoine Lavoisier: The Frenchman who transformed chemistry forever • RFI English by RFI English: 748 views (http://www.youtube.com/watch?v=BjnlS-udeMQ)
Lavoisier and His Wife’s History

Lavoisier’s laboratory, Musée des Arts et Métiers, Paris
(Wiki Image By Edal Anton Lefterov – Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=11621171)
Antoine-Laurent Lavoisier (1743-1794) and Marie-Anne Pierrette Paulze (1758-1836) formed a remarkable scientific partnership central to the Chemical Revolution and the establishment of modern chemistry. Their story is one of intellectual collaboration, personal devotion, and, ultimately, tragedy within the context of the French Revolution.
- Antoine-Laurent Lavoisier: The Scientist
- Background: Born into a wealthy Parisian family, Lavoisier received a broad education, including law, but developed a strong passion for science.
- Early Career: He began his scientific career with work in geology, mineralogy, and public health. He was elected to the prestigious Academy of Sciences in 1768.
- Ferme Générale: He purchased a share in the Ferme générale, a private tax-collection company. This gave him the financial resources to fund his research, but it would later prove fatal.
- The Chemical Revolution: Lavoisier’s most significant contributions were in chemistry:
- Overthrowing Phlogiston: He disproved the phlogiston theory of combustion, demonstrating that combustion involves the combination of a substance with oxygen.
- Law of Conservation of Mass: He established that mass is conserved in chemical reactions.
- Identifying and Naming Elements: He identified and named oxygen and hydrogen and clarified their roles in combustion and water formation.
- Systematic Nomenclature: He developed a new, systematic way of naming chemical compounds.
- Traité Élémentaire de Chimie (1789): His influential textbook presented new theories and nomenclature.
- Quantitative Approach: Lavoisier’s work was characterized by meticulous quantitative experimentation. He used precise instruments and careful measurements to support his theories, a major departure from earlier chemistry’s more qualitative and speculative approaches.
- Marie-Anne Pierrette Paulze: The Collaborator
- Background: Born into a wealthy family, she received a good education, which was unusual for a woman of her time.
- Marriage to Lavoisier (1771): She married Antoine Lavoisier at 13 (a common practice among aristocratic families at the time). The marriage was arranged but developed into a strong intellectual and personal partnership.
- Scientific Training: She received scientific training from Antoine and his colleagues, learning chemistry, mathematics, and drawing.
- Crucial Contributions:
- Laboratory Assistant: She worked alongside Antoine in the laboratory, assisting with experiments, taking detailed notes, and making accurate observations.
- Translator: She was fluent in English and Latin (and, of course, French), and she translated important scientific works into French, including Richard Kirwan’s Essay on Phlogiston (which she also critiqued, pointing out errors that helped convince Lavoisier to abandon phlogiston). This allowed Lavoisier to stay informed about scientific developments in other countries.
- Illustrator: She created detailed and accurate drawings of laboratory apparatus and experiments, essential for illustrating Lavoisier’s publications. Her illustrations were far superior to the typical scientific illustrations of the time.
- Scientific Salon: She hosted a scientific salon, bringing together leading scientists and intellectuals to discuss the latest discoveries and theories. This fostered intellectual exchange and helped to disseminate Lavoisier’s ideas.
- Editor and Publisher: After Antoine’s death, she organized and published his final memoirs (Mémoires de Chimie), ensuring the preservation of his work.
- Their Partnership:
- Collaboration: Their relationship was a true scientific collaboration. Marie-Anne was not simply a passive assistant but an active participant in the research process.
- Complementary Skills: Their skills complemented each other. Antoine was a brilliant theorist and experimentalist; Marie-Anne was a meticulous observer, skilled communicator, and organizer.
- Mutual Respect: They profoundly respected each other’s abilities.
- Social Circle: They mixed with some of the greatest minds in Europe.
- The French Revolution and Tragedy:
- Lavoisier’s Involvement: Antoine Lavoisier was involved in the early stages of the French Revolution, supporting moderate reforms.
- Ferme Générale: His association with the Ferme générale (tax collection) made him a target of the increasingly radical revolutionaries.
- Reign of Terror: During the Reign of Terror, the Ferme générale was abolished, and its members were accused of corruption and treason.
- Arrest and Execution (1794): Antoine Lavoisier was arrested, tried, and executed by guillotine on May 8, 1794.
- Marie-Anne’s Efforts: Marie-Anne attempted to defend Antoine and secure his release, but to no avail.
- Marie-Anne’s Later Life:
- Preserving Lavoisier’s Legacy: Marie-Anne worked tirelessly to preserve and publish his work after Antoine’s death. She organized his papers and notes, and she published his final memoirs.
- Continued Scientific Involvement: She continued to host scientific salons and remained connected to the scientific community.
- Remarriage: She remarried (briefly) to Benjamin Thompson (Count Rumford), another prominent scientist, but the marriage was unhappy and ended in separation.
- Death (1836): She died in 1836 at the age of 78, outliving Antoine by over 40 years.
In summary, Antoine Lavoisier is rightly celebrated as the “father of modern chemistry,” but his achievements were significantly aided by the crucial contributions of his wife, Marie-Anne. She was not simply a supportive spouse; she was a scientific partner, translator, illustrator, and editor. Their story is a testament to the power of collaboration and the often-overlooked contributions of women to the history of science. The French Revolution tragically cut off Antoine’s life and work, but Marie-Anne ensured his legacy would endure. Their partnership is one of the most remarkable in the history of science.
Lavoisier, The Quantitative Revolution, and Legacy. Table

Lavoisier (wearing goggles) operates his solar furnace to prevent contamination from combustion products.
(Wiki Image By Antoine Lavoisier – Science History Institute, Public Domain, https://commons.wikimedia.org/w/index.php?curid=64707215)
Let’s create a table summarizing the key aspects of Antoine Lavoisier’s work, focusing on his role in the “Quantitative Revolution” in chemistry and outlining his lasting legacy. This will provide a structured overview of his impact.
| Aspect | Details | Significance |
| The Quantitative Revolution | Emphasis on Precise Measurement: Lavoisier consistently used the best available balances and other instruments to make precise measurements of the masses and volumes of reactants and products in chemical reactions. <br> – Closed Systems: He performed many experiments in sealed vessels to prevent the escape or entry of gases, allowing for accurate mass measurements. <br> – Systematic Approach: He conducted a systematic series of experiments, carefully varying conditions and observing the results. <br> – Mathematical Analysis: He used mathematical analysis to interpret his experimental data and to formulate his theories. | – Transformed chemistry from a qualitative to a quantitative science. This was his most fundamental contribution. <br> – Enabled the formulation of the Law of Conservation of Mass. <br> – Allowed for accurate determination of the composition of substances. <br> – Set a new standard for scientific rigor in chemistry. |
| Overthrow of Phlogiston Theory | – Phlogiston Theory (Prevailing Theory): Combustion was believed to involve the release of a hypothetical substance called “phlogiston.” <br> – Lavoisier’s Experiments: His experiments on calcination (heating metals in air) and combustion directly contradicted the phlogiston theory. He showed that substances gain weight when they burn, combining with oxygen. | – Demonstrated that the phlogiston theory was incorrect. <br> – Established the oxygen theory of combustion as the correct explanation. <br> – A major turning point in the history of chemistry. |
| Key Discoveries and Concepts | – Oxygen: Identified and named oxygen; demonstrated its role in combustion, respiration, and rusting. <br> – Hydrogen: Identified and named hydrogen; showed that it combines with oxygen to form water. <br> – Water as a Compound: Proved that water is a compound of hydrogen and oxygen, not an element. <br> – Law of Conservation of Mass: Rigorously established that mass is neither created nor destroyed in chemical reactions. | – Fundamental discoveries about the nature of matter and chemical reactions. <br> – Provided the basis for understanding chemical composition and stoichiometry. |
| Chemical Nomenclature | – ** Méthode de Nomenclature Chimique (1787):** Developed (with collaborators) a new, systematic system of chemical nomenclature based on the composition of compounds. | – Replaced the older, inconsistent, and often confusing terminology. <br> – Created a clear and logical language for chemistry, facilitating communication and understanding. <br> – The basis for the system still used today. |
| Textbook and Dissemination | – Traité Élémentaire de Chimie (Elements of Chemistry) (1789): Published a landmark textbook that presented his new theories and nomenclature clearly and organized. | – Revolutionized chemical education. <br> – Disseminated his ideas throughout Europe and beyond, leading to the widespread acceptance of the “new chemistry.” |
| Legacy | – “Father of Modern Chemistry”: Universally recognized for his transformative contributions. <br> – Quantitative Chemistry: Established chemistry as a quantitative science. <br> – Law of Conservation of Mass: A fundamental principle of chemistry and physics. <br> – Oxygen Theory of Combustion: The foundation of our understanding of combustion. <br> – Modern Chemical Nomenclature: His system is the basis for the current nomenclature. <br> – Influence on Other Sciences: His work had a significant impact on biology (respiration) and physics (heat). <br> – Scientific Method: Emphasized the role of a controlled experiment. | – His work marked a definitive break from alchemy and the older, qualitative approaches to chemistry. <br> – He laid the foundations for all subsequent developments in modern chemistry. <br> – His emphasis on quantitative experimentation and clear communication set a new standard for scientific inquiry. |
Export to Sheets
Key Takeaways:
- Quantitative Approach: Lavoisier’s focus on precise measurement and quantitative analysis was revolutionary.
- Experimental Evidence: He used carefully designed experiments to test his hypotheses and to disprove the prevailing phlogiston theory.
- Systematic Thinking: He developed a systematic approach to chemistry, including a new nomenclature system.
- Communication: He recognized the importance of clear communication and presented his work in a systematic and accessible way.
- Lasting Impact: He did not simply make one discovery but changed the fundamental way chemistry was done.
This table provides a comprehensive overview of Antoine Lavoisier’s contributions to chemistry, highlighting his role in the “Quantitative Revolution” and his lasting legacy. It emphasizes the key elements of his work: his experimental methods, groundbreaking discoveries, new system of nomenclature, and dissemination of his ideas through his influential textbook. The format allows for a clear understanding of the interconnectedness of his achievements and their profound impact on the development of modern chemistry.
Lavoisier Quantitative Experimentation. Table
Let’s focus specifically on Antoine Lavoisier’s use of quantitative experimentation, a defining characteristic of his work that revolutionized chemistry. This table will detail key examples of his experiments, highlighting the quantitative aspects (measurements, data), observations, and conclusions, demonstrating how this approach led to his breakthroughs.
| Experiment | Quantitative Methods Used | Key Quantitative Observations | Conclusions Drawn | Significance |
| Calcination of Metals (e.g., Tin, Lead) | – Heating metals in sealed vessels with a known volume of air. <br> – Precise measurement of the mass of the metal before and after heating. <br> – Measurement of the volume of air before and after heating. | – The metal gained weight after heating (forming a calx, or oxide). <br> – The volume of air in the vessel decreased. <br> – The total mass of the system (vessel + metal + air) remained constant. | – Combustion is not the release of phlogiston (which would have caused weight loss). <br> – Metals combine with a component of air during calcination. <br> – Confirms the Law of Conservation of Mass. | – Overthrew the phlogiston theory. <br> – Established the importance of precise measurement in chemistry. <br> – Provided key evidence for the Law of Conservation of Mass. |
| Heating of Mercury Calx (Mercuric Oxide) | – Heating mercury calx (HgO) in a closed system. <br> – Collecting and measuring the volume of the gas released. <br> – Measuring the mass of the mercury calx before and after heating and the mass of the gas produced. | – Mercury calx lost weight upon heating. <br> – A gas that supported combustion better than ordinary air was released. <br> – The weight loss of the calx was equal to the weight of the gas released. | – The gas released is the same one absorbed during calcination (oxygen). <br> – Combustion involves a combination with this gas (oxygen), not release of phlogiston. <br> – Calcination is a reversible process. | – Identified and isolated oxygen. <br> – Further refuted the phlogiston theory. <br> – Demonstrated the reversible nature of chemical reactions. |
| Combustion of Phosphorus and Sulfur | – Burning phosphorus and sulfur in a known volume of air. <br> – Measuring the weight of the phosphorus/sulfur before and after burning. <br> – Observing the absorption of air. | – Phosphorus and sulfur gained weight when burned. <br> – Air was absorbed during the process. <br> – The products were acidic. | – Combustion is the combination of a substance with oxygen. <br> – (Incorrectly) concluded that oxygen was a component of all acids. | – Extended the oxygen theory of combustion beyond metals. |
| Decomposition and Synthesis of Water | – Decomposing water into hydrogen and oxygen using heated iron. <br> – Synthesizing water by burning hydrogen in oxygen. <br> – Careful measurement of the volumes and weights of gases involved. | – Water could be broken down into two distinct gases (hydrogen and oxygen). <br> – These two gases could be recombined to form water. | – Water is a compound of hydrogen and oxygen, not an element. | – Overthrew the ancient belief that water was an element. <br> – Demonstrated the power of quantitative analysis to determine the composition of substances. |
| Respiration Experiments | – Measuring the volume of oxygen consumed and carbon dioxide produced by animals (guinea pigs) in closed containers. <br> – Comparing this to the changes observed during the burning of charcoal. | – Animals consume oxygen and produce carbon dioxide, similar to combustion. <br> – Respiration generates heat. | – Respiration is a slow form of combustion, providing energy for living organisms. <br> – Linked chemical processes to vital biological functions. |
Export to Sheets
Key Principles Illustrated:
- Closed Systems: Lavoisier’s use of sealed containers was crucial for accurately measuring changes in mass and volume, preventing the escape or entry of gases.
- Precise Measurement: He used the best available balances and other instruments to make precise mass and volume measurements, allowing him to draw quantitative conclusions.
- Conservation of Mass: His experiments consistently demonstrated the conservation of mass in chemical reactions, a fundamental principle.
- Quantitative Analysis: He used quantitative data to disprove the phlogiston theory and support his oxygen combustion theory.
- Control: By performing experiments with different starting conditions and having consistent results.
This table highlights Lavoisier’s revolutionary quantitative experimental approach. He didn’t just observe chemical reactions; he measured them and used these measurements to develop and test his theories. This emphasis on quantification was the key to his success in overthrowing the phlogiston theory and establishing the foundations of modern chemistry. The table format clearly shows the link between his methods, observations, and groundbreaking conclusions.
Lavoisier Law of Conservation of Mass. Table
Let’s focus specifically on Antoine Lavoisier’s experiments and reasoning that led to the establishment of the Law of Conservation of Mass, presented in a table format. This will highlight the quantitative nature of his work and how it contrasted with the prevailing phlogiston theory.
| Experiment/Observation | Phlogiston Theory Prediction (Incorrect) | Lavoisier’s Quantitative Experiment and Observation | Lavoisier’s Conclusion (Law of Conservation of Mass) | Significance |
| Calcination of Metals (e.g., Tin, Lead) | – Metal heated in air releases phlogiston. <br> – Calx (residue) should weigh less than the original metal because phlogiston has been lost. | – Metal heated in a sealed vessel with a known amount of air. <br> – Carefully measured the mass of the metal, vessel, and air before and after heating. <br> – Observed that the metal gained weight (forming a calx). <br> – Observed that the vessel’s air volume decreased. <br> – Crucially: The total mass of the sealed system (vessel + metal + air) remained constant. | – Mass is neither created nor destroyed in a chemical reaction; it is conserved. <br> – The metal combines with air (oxygen) components during calcination, increasing its weight. <br> – Phlogiston theory is incorrect. | – Established the Law of Conservation of Mass as a fundamental principle of chemistry. <br> – Showed that combustion is a combination with oxygen, not the release of phlogiston. <br> – Emphasized the importance of quantitative measurements. |
| Heating of Mercury Calx (Mercuric Oxide) | – Phlogiston would be released. | – Mercury calx (HgO) heated in a closed system. <br> – Collected and measured the volume of the gas released. <br> – Measured the mass of the mercury calx before and after heating and the mass of the gas. | – The gas released is the same gas absorbed during calcination (oxygen). <br> – The mass loss of the calx equals the mass of the gas released. <br> – Mass is conserved. | – Confirmed that the process is reversible and that a specific air component (oxygen) is involved. <br> – Further evidence against phlogiston. |
| Combustion of Phosphorus and Sulfur | – Burning involved the release of Phlogiston. | – Phosphorus/sulfur burned in a known volume of air. <br> – Measured the weight of phosphorus/sulfur before and after burning. | -The weight increased. <br> – Mass is conserved. | – Combustion is a combination. |
| Decomposition and Synthesis of Water | – Water is considered a single element. | – Measured amounts of Water decomposed into hydrogen and oxygen. <br> – Hydrogen and oxygen recombined to form water. | – Water could be broken down into two distinct gases (hydrogen and oxygen). <br> – These two gases could be recombined to form water in definite proportions. | – Showed it was a compound. |
Export to Sheets
Key Principles Demonstrated by Lavoisier’s Experiments:
- Closed Systems: Using sealed vessels was essential to prevent the escape or entry of gases, allowing for accurate mass measurements.
- Precise Measurement: Lavoisier used the best available balances to measure mass with the greatest possible accuracy. This quantitative approach was revolutionary.
- Systematic Approach: He conducted carefully designed experiments, systematically varying conditions and observing the results.
- Rejection of Phlogiston: His quantitative data directly contradicted the predictions of the phlogiston theory.
- Conservation as a Fundamental Law: He demonstrated that mass is conserved in chemical reactions, a cornerstone of modern chemistry.
This table demonstrates how Lavoisier’s meticulous quantitative experiments, focused on measuring mass changes, led him to establish the Law of Conservation of Mass and overthrow the phlogiston theory. The table contrasts the predictions of the old theory with Lavoisier’s actual observations and conclusions, highlighting the revolutionary nature of his work. It emphasizes that his method (precise measurement in closed systems) and observations led to his breakthroughs.
Lavoisier Combustion and Oxygen. Table
Let’s create a table summarizing Antoine Lavoisier’s key experiments, observations, and conclusions related explicitly to combustion and oxygen. This will separate it from the broader table on the Law of Conservation of Mass (although the two are inherently linked) and focus on his understanding of combustion and oxygen’s role.
| Experiment/Observation | Lavoisier’s Setup and Procedure | Key Observations | Lavoisier’s Conclusions | Significance |
| Calcination of Metals (Tin, Lead) | Heated metals in sealed vessels with a known volume of air; carefully measured masses of reactants and products. | – Metal gained weight (forming a calx/oxide). <br> – Volume of air in the vessel decreased. <br> – Total mass of the system remained constant. | – Combustion is not the release of phlogiston (which would cause weight loss). <br> – Metals combine with a component of air during calcination. | – Overthrew the phlogiston theory. <br> – Demonstrated that combustion involves a combination with something in the air. |
| Heating Mercury Calx (Mercuric Oxide) | Heated mercury calx (HgO) in a closed system; collected and measured the volume and mass of the gas released. | – Mercury calx lost weight. <br> – A gas that supported combustion more vigorously than ordinary air was released. <br> – Mass loss of calx = mass of gas released. | – The gas released is the same gas absorbed during calcination. <br> – This gas (later named “oxygen”) is essential for combustion. | – Identified and isolated oxygen (though he didn’t initially call it that). <br> – Showed that combustion is a reversible process. |
| Analysis of Air | Various methods (including the mercury calx experiments) were used to analyze the composition of air. | – Air is a mixture of gases, primarily <br> – “Eminently respirable air” (oxygen) which supports combustion and respiration. <br> – An inert gas (nitrogen) that does not. | – Identified oxygen as the active component of air in combustion. <br> – Showed that air is not a single element (as previously believed). | |
| Combustion of Phosphorus and Sulfur | Burned phosphorus and sulfur in a known volume of air; measured weight changes and observed the products. | – Phosphorus and sulfur gained weight when burned. <br> – The products formed were acidic when dissolved in water. | – Combustion is the combination of a substance with oxygen. <br> – (Incorrectly) concluded that oxygen was a component of all acids (hence the name “oxygen”). | – Extended the oxygen theory of combustion beyond metals. <br> – (Incorrectly) linked oxygen to acidity, but this productive error led to further research. |
| Respiration Experiments | Measured the volume of oxygen consumed and carbon dioxide produced by animals (guinea pigs) in closed containers; compare this to the burning of charcoal. | – Animals consume oxygen and produce carbon dioxide, similar to combustion. <br> – Respiration generates heat. | – Respiration is a slow form of combustion, providing energy for living organisms. | – Connected chemical processes (combustion) to vital biological functions (respiration). |
Export to Sheets
Key Principles Demonstrated:
- Quantitative Approach: Lavoisier’s precise mass and volume measurements were crucial to his discoveries.
- Closed Systems: Using sealed vessels was essential to prevent the escape or entry of gases and demonstrate mass conservation.
- Oxygen as the Key: He identified oxygen as the key element involved in combustion, respiration, and forming many acids (though this last point was later refined).
- Rejection of Phlogiston: His experiments and conclusions directly contradicted and disproved the phlogiston theory.
This table focuses on Lavoisier’s work on combustion and oxygen, showing how his experiments led to his revolutionary understanding of these processes. It separates the experiments, methods, observations, and conclusions, making the logic of his discoveries clear. The table emphasizes his work’s quantitative nature and oxygen’s crucial role in his new chemical theory.
Lavoisier Systematic and Chemical Nomenclature. Table
Let’s detail Antoine Lavoisier’s contributions to systematic chemical nomenclature in a table format. This will contrast the older, often confusing system with his new, rational approach and highlight his work’s key principles and impact. This work was done in collaboration with other prominent chemists of the time (de Morveau, Berthollet, and Fourcroy), culminating in the publication of Méthode de Nomenclature Chimique (Method of Chemical Nomenclature) in 1787.
| Feature | Traditional (Pre-Lavoisier) Nomenclature | Lavoisier’s New System (and Modern System) | Significance |
| Naming Principles | – Often based on arbitrary properties (appearance, origin, supposed alchemical relationships). <br> – Inconsistent and unsystematic. <br> – Many different names for the same substance. | – Based on chemical composition. <br> – Systematic and consistent. <br> – Names reflect the elements present in the compound. <br> – Prefixes and suffixes indicate proportions (though this was further refined later). | – Clarity and Precision: Made chemical communication more transparent and precise. <br> – Universality: Provided a universal system that chemists could understand regardless of their language. <br> – Foundation for Modern Nomenclature: The basis for the system still used today. |
| Examples | – “Oil of vitriol” <br> – “Butter of antimony” <br> – “Flowers of zinc” <br> – “Blue vitriol” <br> -“Calx” | – “Sulfuric acid” (H₂SO₄) <br> – “Antimony trichloride” (SbCl₃) <br> – “Zinc oxide” (ZnO) <br> – “Copper(II) sulfate” (CuSO₄) <br> -“Oxide” | – Illustrates the confusing and often metaphorical nature of the old names. <br> – Shows how the new names directly relate to the chemical composition. |
| Elements | – Many substances considered elements were compounds (e.g., water, lime). <br> – Elements were often associated with alchemical symbols and concepts. | – Defined elements as substances that cannot be broken down into simpler substances by chemical means. <br> – Provided a clear list of known elements. | – Established a clear definition of an element, a fundamental concept in chemistry. <br> – Helped to distinguish between elements and compounds. |
| Compounds | – Names often gave no indication of composition. | – Names indicated the elements present and, to some extent, their proportions (e.g., “sulfurous acid” vs. “sulfuric acid” – though the precise understanding of stoichiometry came later). | – Made it possible to deduce the composition of a compound from its name. <br> – Facilitated the understanding of chemical reactions. |
| Acids, Bases, and Salts | – Inconsistent and often confusing terminology. | – Classified substances based on their properties and composition. <br> – (Incorrectly) believed that all acids contained oxygen (hence the name). <br> – Developed a more systematic way of naming salts. | – Provided a more organized and rational classification of chemical substances. <br> – (Although the oxygen theory of acids was wrong, it stimulated further research.) |
| Impact on Chemical Theory | – The old nomenclature was tied to outdated theories (like phlogiston). | – The new nomenclature was directly linked to Lavoisier’s new chemical theories (oxygen theory of combustion, law of conservation of mass). | – Facilitated the acceptance of the new chemistry. A clear and logical naming system made it easier for chemists to understand and adopt Lavoisier’s revolutionary ideas. |
| Publication | – | Méthode de Nomenclature Chimique (1787) | – |
Export to Sheets
Key Principles of Lavoisier’s System:
- Composition-Based: Names should reflect the chemical composition of the substance.
- Systematic: A consistent set of rules should be used to generate names.
- Unambiguous: Each substance should have a unique and unambiguous name.
- International: The system should be understandable to chemists of all nationalities (Latin was used as the basis for many names).
This table contrasts the old, unsystematic nomenclature with Lavoisier’s new, rational system, highlighting the key principles and the profound impact of this change on the development of chemistry. It demonstrates how a seemingly “technical” detail – how we name chemical compounds – can be crucial for scientific progress. The new nomenclature was not just a matter of convenience but integral to the Chemical Revolution, providing a clear and consistent language for the new chemistry.
Lavoisier Collaboration, Scientific Method, Elements of Chemistry (Textbook). Table
Antoine Lavoisier’s contributions were pivotal in the transformation of chemistry into a modern, quantitative science. Here’s a table summarizing his key contributions, focusing on collaboration, the scientific method, and his influential textbook:
Antoine Lavoisier: Key Contributions
| Contribution | Description | Significance |
| Collaboration | * Worked with his wife, Marie-Anne Lavoisier, who translated scientific works, illustrated his experiments, and participated in his research. <br> * Collaborated with other scientists of his time. | * Emphasized the importance of teamwork in scientific progress. <br> * Marie-Anne’s contributions were invaluable in disseminating Lavoisier’s findings. |
| Scientific Method | * Emphasized precise measurements and quantitative analysis in chemical experiments. <br> * Systematically used experimentation to test hypotheses. <br> * Stressed the importance of careful observation and logical deduction. | * Helped to establish chemistry as a rigorous, quantitative science. <br> * His work exemplified the application of the scientific method to chemical investigations. <br> * His emphasis on precise measurement lead to the discoverey of the law of conservation of mass. |
| Elements of Chemistry (“Traité Élémentaire de Chimie”) | * Published in 1789, it was the first modern chemistry textbook. <br> * Defined elements as substances that could not be broken down further by chemical analysis. <br> * Introduced a new system of chemical nomenclature. <br> * Organized known elements into a table. | * Revolutionized chemical education and standardized chemical terminology. <br> * Helped to disseminate his new chemistry and establish it as the dominant scientific view. <br> * Considered one of the most influential books in the history of chemistry. |
Export to Sheets
Key Points:
- A meticulous approach to experimentation and a strong emphasis on quantitative data characterized Lavoisier’s work.
- His textbook, “Traité Élémentaire de Chimie,” played a crucial role in establishing the foundations of modern chemistry.
- His wife’s contributions were extremely valuable to his overall work.
John Dalton (1766-1844) – Atomic Theory

Dalton by Thomas Phillips, 1835
(Wiki Image By Thomas Phillips – National Portrait Gallery, London, Public Domain, https://commons.wikimedia.org/w/index.php?curid=11727058)
John Dalton Quotes Table
John Dalton’s contributions to science were immense, and while he may not be as widely known for pithy quotes as some other historical figures, he did leave behind statements that reveal his approach to science and his key ideas. Here’s a table with some of his notable quotes:
John Dalton Quotes
| Quote | Theme/Context |
| “Atoms cannot be seen, but we infer their existence and properties from the ways in which substances behave.” | Atomic theory, the nature of scientific evidence |
| “In science, the credit goes to the man who convinces the world, not to the man to whom the idea first occurs.” | Scientific recognition, the importance of proving theories |
| “The laws of chemical combination are the best evidence of the existence of atoms.” | Evidence for atomic theory, chemical laws |
| “It may be remarked that if we cannot always discover the causes of things, we can sometimes discover the laws to which they are subject.” | Scientific principles and the discovery of natural laws. |
| “Chemical analysis and synthesis go no farther than to the separation of particles one from another and to their reunion. No new creation or destruction of matter is within the reach of chemical agency.” | Chemical reactions and the law of conservation of mass. |
| “It’s the right idea, but not the right time.” | This is a more general quote that can be applied to many situations. |
| “If I have succeeded better than many who surround me, it has been chiefly – may I say almost solely – from universal assiduity.” | This quote shows his dedication to his work. |
Export to Sheets
Key Observations:
- Dalton’s quotes often reflect his focus on empirical evidence and the importance of observation in scientific inquiry.
- His statements about atoms and chemical combinations highlight his groundbreaking work in atomic theory.
- His quote on scientific credit displays an understanding of how scientific advancement occurs.
I hope this table is helpful.
John Dalton YouTube Video
- Dalton’s Atomic Theory by The Organic Chemistry Tutor: 179,274 views (http://www.youtube.com/watch?v=rbmPpgASUAY)
- John Dalton Biography by CloudBio: 71,200 views (http://www.youtube.com/watch?v=sDoH5fPTOfo)
- How John Dalton’s meteorological studies led to the discovery of atoms by Science History Institute: 59,047 views (http://www.youtube.com/watch?v=d2WWgTGJsIw)
- John Dalton: First Scientist to Study Color Blindness | Biography by Biography: 41,978 views (http://www.youtube.com/watch?v=CGvS3qiUDbY)
- Biography of John Dalton || first atomic model by The Vodcast: 4,505 views (http://www.youtube.com/watch?v=RhTBCPhS-qU)
John Dalton History
Color Blindness (Daltonism)
(Wiki Image By Shinobu Ishihara – File:Ishihara 9.png, Public Domain, https://commons.wikimedia.org/w/index.php?curid=104034287)
John Dalton (1766-1844) was an English chemist, physicist, and meteorologist best known for pioneering modern atomic theory development. His work marked a fundamental shift in understanding matter, laying the groundwork for modern chemistry and physics.
- Early Life and Education (1766-1793):
- Humble Beginnings: Born on September 6, 1766, in Eaglesfield, Cumberland, England, into a modest Quaker family. His father was a weaver. The Quakers, known for emphasizing education and practical skills, likely influenced his approach to science.
- Early Education: He received his early education at a Quaker school in his village. He displayed an exceptional aptitude for learning, particularly in mathematics and science.
- Precocious Teacher: He began teaching at a local school at the remarkably young age of 12, demonstrating his early intellectual abilities.
- Kendal School (1781-1793): Moved to Kendal and became a teacher and a principal at a Quaker boarding school. He taught mathematics, natural philosophy (science), and English grammar.
- Self-Education: Dalton was largely self-taught in science and mathematics. He read widely, conducted his experiments, and engaged in intellectual discussions with other scholars in the area, including John Gough, a blind philosopher and polymath who mentored him.
- Early Interest in Meteorology: He developed a keen interest in meteorology, beginning a lifelong habit of daily weather observations (he kept a weather journal for 57 years). This meticulous record-keeping and observation became a hallmark of his scientific approach.
- Manchester and Scientific Development (1793-1844):
- Move to Manchester (1793): He moved to Manchester, a rapidly growing industrial city, and became a tutor in mathematics and natural philosophy at the Manchester Academy (also known as New College), a dissenting academy (for students who were not members of the Church of England).
- Manchester Literary and Philosophical Society: He joined the Manchester Literary and Philosophical Society, a prominent scientific society, where he presented many scientific papers. This provided him a platform for sharing his ideas and engaging with other scientists.
- Early Scientific Work (pre-atomic theory):
- Meteorology: He continued his meteorological observations and published Meteorological Observations and Essays (1793), his first major scientific work.
- Gas Laws: Studied the behavior of gases, investigating their properties, diffusion, and absorption by liquids. This work laid the groundwork for his later atomic theory. He formulated Dalton’s Law of Partial Pressures (1801), stating that the total pressure of a mixture of gases is equal to the sum of the partial pressures of the individual gases.
- Color Blindness (Daltonism): He published a paper describing color blindness (1794), which he experienced. This condition is sometimes called “Daltonism” in his honor.
- Development of the Atomic Theory (1803-1808):
- The Crucial Insight: Dalton’s key insight was to connect the quantitative relationships observed in chemical reactions (e.g., the Law of Definite Proportions and the Law of Multiple Proportions) with the ancient Greek idea of atoms. He realized these laws could be explained if matter were composed of indivisible atoms of different elements.
- Key Postulates of Dalton’s Atomic Theory:
- All matter is made of atoms, which are indivisible and indestructible. (This was later proven incorrect with the discovery of subatomic particles, but the core concept of atoms remains.)
- All atoms of a given element are identical in mass and properties. (We now know about isotopes, but elements have distinct chemical properties.)
- Compounds are formed by combining two or more different kinds of atoms.
- A chemical reaction is a rearrangement of atoms.
- Law of Multiple Proportions: Dalton’s observation that elements combine in simple, whole-number ratios (e.g., carbon and oxygen can form CO or CO₂, but not CO₁.₅) provided strong experimental evidence for his atomic theory.
- First Table of Atomic Weights: He created the first table of relative atomic weights, assigning a weight to each known element (using hydrogen as the standard, with a weight of 1). While many of his values were inaccurate by modern standards, this was a crucial step in quantifying chemistry.
- A New System of Chemical Philosophy (1808-1827): Dalton published his atomic theory and table of atomic weights in his multi-part book, A New System of Chemical Philosophy. This was a landmark work in the history of chemistry, clearly presenting his revolutionary ideas.
- Later Life and Recognition:
- Gradual Acceptance of Atomic Theory: Dalton’s atomic theory was not immediately accepted by all chemists, but it gradually gained support as more experimental evidence accumulated.
- Continued Research and Teaching: He continued to conduct research (though his later work was less significant than his atomic theory) and to teach in Manchester.
- Honors and Awards: He received numerous honors and awards, including membership in the Royal Society (though he was initially reluctant to accept it).
- Quiet Life: He lived a relatively quiet and unassuming life, devoted to his scientific work. He never married.
- Death and Legacy:
- Death (1844): He died in Manchester on July 27, 1844.
- “Father of Modern Atomic Theory”: He is considered the father of modern atomic theory, the foundation upon which much of modern chemistry and physics is built.
- Revolutionized Chemistry: His theory transformed chemistry from a largely empirical science into one based on a clear theoretical framework.
- Quantitative Approach: He emphasized the importance of quantitative relationships in chemistry.
- Lasting Impact: While some details of his theory have been modified (e.g., atoms are divisible, isotopes exist), the core concept of atoms as the fundamental building blocks of matter remains central to all of science.
Despite humble beginnings and limited formal scientific training, John Dalton’s life story is a remarkable intellectual achievement. His meticulous observations, insightful reasoning, and bold theorizing led to one of history’s most important scientific breakthroughs—the modern atomic theory. He is a prime example of how careful observation of the natural world and willingness to challenge existing ideas can lead to profound scientific advances.
John Dalton, Atomic Theory, and Legacy. Table
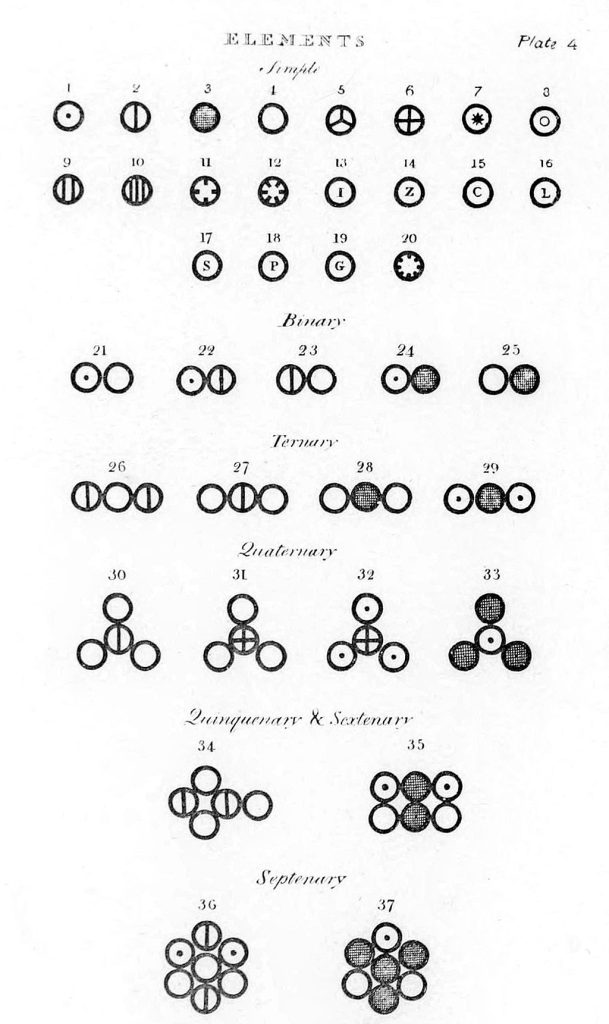
Various atoms and molecules, as depicted in John Dalton’s A New System of Chemical Philosophy (1808)
(Wiki Image By haade – En.wiki (https://archive.org/details/newsystemofchemi01daltuoft/page/n237), Public Domain, https://commons.wikimedia.org/w/index.php?curid=1387026)
Let’s summarize John Dalton’s (1766-1844) life, his development of the atomic theory, and his lasting legacy in a structured table format. This will present the key aspects of his work and its significance.
| Aspect | Details |
| Early Life and Education (1766-1793) | – Born in Eaglesfield, Cumberland, England (1766), to a Quaker family. <br> – Early education at a Quaker school; began teaching at age 12. <br> – Largely self-taught in science and mathematics. |
| Manchester Period (1793-1844) | – Moved to Manchester (1793); tutor at the New College. <br> – Joined the Manchester Literary and Philosophical Society (a scientific society). |
| Early Scientific Work | – Meteorology: Kept a daily weather journal for 57 years; meticulous observations. <br> – Gas Laws: Studied the behavior of gases; formulated Dalton’s Law of Partial Pressures (1801). <br> – Color Blindness: One of the first scientific descriptions of color blindness (“Daltonism”). |
| Development of Atomic Theory (1803-1808) | – Key Postulates: <br> 1. All matter is made of indivisible and indestructible atoms. <br> 2. Atoms of a given element are identical in mass and properties. <br> 3. Combinations of atoms of different elements form compounds. <br> 4. A chemical reaction is a rearrangement of atoms. <br> – Law of Multiple Proportions: Observed that elements combine in simple, whole-number ratios (e.g., CO vs. CO₂), providing evidence for his theory. <br> – First Table of Atomic Weights: Created the first table of relative atomic weights (though many values were inaccurate). |
| Publication and Dissemination | – A New System of Chemical Philosophy (1808-1827): He published his atomic theory and table of atomic weights in this multi-part work. <br> – Lectures: Presented his ideas in Manchester, Edinburgh, and London lectures. |
| Later Career | – Continued teaching and research in Manchester. <br> – Received increasing recognition for his atomic theory. |
| Legacy | – Father of Modern Atomic Theory: His atomic theory is the foundation of modern chemistry. <br> – Quantitative Chemistry: His work emphasized the importance of quantitative relationships in chemistry. <br> – Influence on Other Scientists: Inspired and influenced generations of chemists. <br> – Conceptual Framework: Provided the conceptual framework for understanding chemical reactions and the composition of matter. <br> – Lasting Impact: While some details of his theory have been modified (e.g., atoms are divisible, isotopes exist), the core concept of atoms as the building blocks of matter remains central to chemistry. |
| Key Experiments | Gas studies, the combination of elements. |
Export to Sheets
Key Elements of Dalton’s Atomic Theory:
- Atoms as Indivisible Particles: The (now modified) idea that matter is composed of tiny, indivisible particles called atoms.
- Elements and Atoms: The concept that each element is composed of its unique type of atom.
- Compounds and Combinations: The idea that compounds are formed by combining atoms of different elements in fixed ratios.
- Chemical Reactions as Rearrangements: The understanding that chemical reactions involve the rearrangement of atoms, not their creation or destruction.
- Relative Atomic Weights: The attempt to determine the relative weights of atoms, providing a quantitative basis for chemical calculations.
Why Dalton’s Theory Was Revolutionary:
- Shift from Qualitative to Quantitative: It moved chemistry from a largely qualitative and descriptive science to a more quantitative one.
- Explanatory Power: It provides a simple and elegant explanation for various chemical phenomena (laws of definite and multiple proportions).
- Predictive Power: It allowed for predictions about the behavior of chemical substances.
- Foundation for Future Research: It laid the foundation for much 19th-century chemistry and later developments in atomic physics.
This table provides a clear and structured overview of John Dalton’s life, his development of the atomic theory, and his lasting legacy on chemistry. It highlights the key postulates of his theory, the evidence he used to support it, and its profound impact on the field. The format allows for an easy understanding of his ideas’ progression and significance.
Dalton Atoms as Indivisible Particles: The (now modified) idea that matter is composed of tiny, indivisible particles called atoms. Table
John Dalton’s atomic theory was a groundbreaking concept in chemistry, though it has undergone modifications as our understanding of atomic structure has deepened. Here’s a breakdown in a table format, highlighting the original postulates and their modern modifications:
Dalton’s Atomic Theory: Original Postulates vs. Modern Understanding
| Original Postulate | Modern Understanding | Notes |
| All matter is composed of extremely small particles called atoms. | True. | The fundamental idea that matter is made of atoms remains correct. |
| Atoms of a given element are identical in size, mass, and other properties. | Atoms of the same element can have different masses (isotopes). | The discovery of isotopes revealed that atoms of the same element can have varying numbers of neutrons, leading to different atomic masses. |
| Atoms cannot be subdivided, created, or destroyed. | Atoms can be subdivided into subatomic particles (protons, neutrons, electrons). Nuclear reactions can change atoms. | The discovery of subatomic particles and nuclear reactions showed that atoms are not indivisible and can be transformed. |
| Atoms of different elements combine in simple whole-number ratios to form chemical compounds. | True. | This principle remains a cornerstone of stoichiometry and chemical combination. |
| In chemical reactions, atoms are combined, separated, or rearranged. | True. | This concept is fundamental to understanding how chemical reactions occur. |
Export to Sheets
Key takeaways:
- Dalton’s theory was a pivotal step in the development of atomic theory.
- While some of his postulates have been modified, his core idea that matter is composed of atoms remains valid.
- Modern atomic theory accounts for subatomic particles, isotopes, and nuclear reactions.
I hope this helps.
Dalton Elements and Atoms: Each element is composed of a unique type of atom. Table
It’s important to present Dalton’s concepts accurately while acknowledging modern atomic theory advancements. Here’s a table that aims to clearly illustrate the core of Dalton’s ideas regarding elements and atoms and how they relate to what we understand today:
Dalton’s Atomic Theory: Elements and Atoms
| Concept | Dalton’s Original Idea | Modern Scientific Understanding |
| Nature of Elements | Each element is composed of extremely small particles called atoms. | The concept of elements as fundamental substances remains. |
| Atom Uniqueness | Atoms of a given element are identical in size, mass, and other properties. Atoms of different elements differ in size, mass, and other properties. | Atoms of an element share a defining number of protons (atomic number), but can vary in mass (isotopes). Atoms of different elements always differ in the number of protons. |
| Atom Indivisibility | Atoms cannot be subdivided, created, or destroyed. | Atoms are composed of subatomic particles (protons, neutrons, electrons) and can be transformed in nuclear reactions. |
| Element Definition | An element is defined by the type of atom it contains. | An element is defined by the number of protons within the atom’s nucleus. |
Export to Sheets
Key Points to Remember:
- Foundation:
- Dalton’s theory was a crucial step in the development of modern chemistry.
- Modifications:
- The discovery of subatomic particles and isotopes led to significant modifications of Dalton’s original postulates.
- Core Principle:
- The idea that elements are composed of atoms remains a fundamental principle.
- Modern Refinements:
- The modern periodic table and the concept of atomic number are direct products of the refinement of Dalton’s original theory.
I hope this table is helpful.
Dalton Compounds and Combinations: The idea that compounds are formed by combining atoms of different elements in fixed ratios. Table
You’re right to highlight the significance of Dalton’s concept of compounds and combinations. It was a major leap forward in understanding how chemical substances are formed. Here’s a table summarizing Dalton’s idea and its modern interpretation:
Dalton’s Atomic Theory: Compounds and Combinations
| Concept | Dalton’s Original Idea | Modern Scientific Understanding |
| Compound Formation | Compounds are formed when atoms of different elements combine. | This fundamental idea remains true. |
| Fixed Ratios | Atoms combine in simple, whole-number ratios to form compounds. | This principle of fixed ratios is still a cornerstone of stoichiometry. |
| Chemical Formulas | While Dalton did not use our current formula methods, his theory laid the groundwork for them. | Chemical formulas represent the fixed ratios of atoms in compounds. Molecular formulas show the actual number of atoms, and empirical formulas show the simplest whole-number ratio. |
| Chemical Reactions | In chemical reactions, atoms are combined, separated, or rearranged. | This remains a core concept; however, we now understand that electron sharing and transfer are the mechanisms behind these rearrangements. |
| Molecular Structure | Dalton did not understand molecular structure. | We now understand that compounds have specific molecular structures that determine their properties. |
Export to Sheets
Key Points:
- Law of Definite Proportions:
- Dalton’s theory explained the law of definite proportions, which states that a chemical compound always contains the same elements in the same proportions by mass.
- Stoichiometry:
- His concept of fixed ratios is the basis of stoichiometry, the branch of chemistry that deals with the quantitative relationships between reactants and products in chemical reactions.
- Chemical Formulas:
- While Dalton did not use the symbolic formulas we use today, his work paved the way for the development of chemical formulas.
- Modern Refinements:
- Modern chemistry has expanded upon Dalton’s ideas by explaining the mechanisms of chemical bonding and the three-dimensional structures of molecules.
Dalton Chemical Reactions as Rearrangements: The understanding that chemical reactions involve the rearrangement of atoms, not their creation or destruction. Table
You’ve hit on another key aspect of Dalton’s atomic theory: his explanation of chemical reactions. Here’s a table that breaks down his concept and its modern interpretation:
Dalton’s Atomic Theory: Chemical Reactions as Rearrangements
| Concept | Dalton’s Original Idea | Modern Scientific Understanding |
| Atom Conservation | Atoms are neither created nor destroyed in chemical reactions. | This fundamental principle, known as the law of conservation of mass, remains a cornerstone of chemistry. |
| Reaction Mechanism | Chemical reactions involve the combination, separation, or rearrangement of atoms. | This is correct, but we now understand the role of electron transfer and sharing in forming and breaking chemical bonds. |
| Mass Conservation | Because atoms are neither created nor destroyed, the total mass of the reactants is equal to the total mass of the products. | This is the law of conservation of mass and is still accepted. |
| Nuclear Reactions | Dalton did not know about nuclear reactions. | We now know that nuclear reactions change the nucleus of atoms and, therefore, the elements themselves. |
| Bonding | Dalton did not understand the nature of chemical bonds. | We now understand that chemical bonds hold atoms together in molecules and compounds, involving the interaction of electrons. |
Export to Sheets
Key Points:
- Law of Conservation of Mass:
- Dalton’s theory provided a clear explanation for the law of conservation of mass, which states that mass is neither created nor destroyed in a chemical reaction.
- Rearrangement, Not Transformation (in Chemical Reactions):
- He emphasized that chemical reactions involve the rearrangement of atoms, not their creation or destruction. This distinction is crucial for understanding chemical processes.
- Electron Interactions:
- Modern chemistry has expanded upon Dalton’s idea by explaining that chemical reactions involve the interactions of electrons, which lead to the formation and breaking of chemical bonds.
- Nuclear Chemistry Exception:
- It is very important to note that Nuclear chemistry is an exception to this rule. Nuclear reactions DO change the atoms themselves.
Dalton Relative Atomic Weights: The attempt to determine the relative weights of atoms, providing a quantitative basis for chemical calculations. Table
You’re right to point out the crucial role of Dalton’s attempts to establish relative atomic weights. While his initial values were often inaccurate, his work laid the foundation for quantitative chemistry. Here’s a table outlining his efforts and their modern context:
Dalton’s Atomic Theory: Relative Atomic Weights
| Concept | Dalton’s Original Idea | Modern Scientific Understanding |
| Relative Weights | Atoms of different elements have different weights, and these weights are relative to a standard (initially hydrogen). | The concept of relative atomic masses is fundamental, but the standard has changed. |
| Determination Method | Dalton attempted to determine relative weights by analyzing the composition of compounds, assuming simple formulas. | Modern methods use mass spectrometry, which provides highly accurate measurements of atomic masses. |
| Initial Standard | Hydrogen was initially used as the standard, with a relative weight of 1. | Carbon-12 is now the international standard, with a relative atomic mass of exactly 12 atomic mass units (u). |
| Accuracy | Dalton’s initial values were often inaccurate due to incorrect assumptions about compound formulas. | Modern relative atomic masses are highly precise. |
| Importance | Dalton’s work provided the first quantitative basis for comparing the masses of atoms, enabling chemical calculations. | Relative atomic masses are essential for stoichiometry, chemical formulas, and many other areas of chemistry. |
| Atomic Mass Unit (AMU) | Dalton did not use the amu. | The atomic mass unit (amu or u) expresses relative atomic masses. |
Export to Sheets
Key Points:
- Pioneering Effort:
- Dalton’s attempt to establish relative atomic weights was a pioneering effort that marked a significant step forward in chemistry.
- Foundation for Stoichiometry:
- His work provided the basis for stoichiometry, enabling chemists to calculate the amounts of reactants and products involved in chemical reactions.
- Modern Mass Spectrometry:
- Modern mass spectrometry has revolutionized the determination of atomic masses, providing highly accurate values.
- Carbon-12 Standard:
- The shift to carbon-12 as the standard has provided a more accurate and consistent basis for relative atomic masses.
- Atomic weight vs Atomic Mass: It is important to note the difference between atomic weight and atomic mass. Atomic mass is the mass of an atom, while atomic weight is the weighted average of the atomic masses of an element’s naturally occurring isotopes.
Jöns Jacob Berzelius (1779-1848) – Standardization and Systematization

Jöns Jacob Berzelius
(Wiki Image By Johan Way – Vetenskapsakademiens porträttsamling, Public Domain, https://commons.wikimedia.org/w/index.php?curid=91568707)
Berzelius Quotes Table
Jöns Jacob Berzelius was a highly influential Swedish chemist whose work significantly shaped the development of modern chemistry. Here’s a table presenting some of his notable quotes, reflecting his contributions and perspectives:
Jöns Jacob Berzelius Quotes
| Quote | Theme/Context |
| “Every chemical combination is wholly and solely dependent on two opposing forces, positive and negative electricity, and every chemical compound must be composed of two parts combined by the agency of their electrochemical reaction since there is no third force.” | Electrochemical theory, the nature of the chemical combination. This demonstrates his early electrochemical theories. |
| “Chemical signs ought to be letters, for the greater facility of writing, and not to disfigure a printed book … I shall take, therefore, for the chemical sign, the initial letter of the Latin name of each elementary substance.” | Chemical notation, the development of symbolic representation. This shows his contribution to modern chemical symbolism. |
| “A catalyst is a substance that alters the velocity of a chemical reaction without appearing in the final products.” | Catalysis is the definition of a key chemical concept. This, of course, remains the basic definition of a catalyst. |
| “A tidy laboratory means a lazy chemist.” | On the importance of doing laboratory work and not just focusing on the organization. |
| “The devil may write textbooks of chemistry, for every few years, the whole thing changes.” | This quote illustrates the dynamic and ever-evolving nature of chemistry. |
Export to Sheets
Key Observations:
- Berzelius’s quotes highlight his focus on electrochemical theory, chemical notation, and the precise definition of chemical concepts.
- His work laid the groundwork for many fundamental principles in chemistry, and his quotes reflect his dedication to clarity and precision.
- It is very clear that he understood that science is not static and is always changing.
Berzelius YouTube Video
- Berzelius Day – Periodic Table of Videos by Periodic Videos: 67,364 views (http://www.youtube.com/watch?v=wDXUDq2KLM4)
- 21. Berzelius to Liebig and Wöhler (1805-1832) by YaleCourses: 9,688 views (http://www.youtube.com/watch?v=Xo_lkUx2BPU)
- Chemistry Video – Jon Jacob Berzelius by Cody E: 520 views (http://www.youtube.com/watch?v=BEVhfqM4yBo)
- Who was Jöns Jacob Berzelius? by Real Life Top 10s: 421 views (http://www.youtube.com/watch?v=pDPdph9Ub98)
Berzelius History

Illustration of Berzelius (published 1903)
(Wiki Image By Unknown author – Front illustration (page 1) from the book, scanned by Gallica.bnf.fr, Public Domain, https://commons.wikimedia.org/w/index.php?curid=10962423)
Jöns Jacob Berzelius (1779-1848) was a Swedish chemist considered one of the founders of modern chemistry. He made fundamental contributions to chemical notation, stoichiometry, analytical chemistry, and the discovery and isolation of elements. His work helped systematize and quantify chemistry, building upon the foundations of Lavoisier and Dalton.
- Early Life and Education (1779-1810):
- Born in Väversunda, Sweden (1779): Orphaned at a young age, he had a difficult childhood.
- Medical Studies at Uppsala University (1796-1802): Initially studied medicine but became increasingly interested in chemistry. His early research focused on the chemical composition of mineral waters.
- Assistant Professor of Medicine and Pharmacy (1802): Appointed to this position at the Karolinska Institute in Stockholm. This provided him with a laboratory and resources for his chemical research.
- Early Electrochemical Experiments: Conducted experiments with Alessandro Volta’s newly invented voltaic pile (battery), investigating the effects of electricity on chemical compounds.
- Key Contributions to Chemistry (1807-1848):
- Modern Chemical Notation (1811-1819):
- Problem: Existing chemical notation was cumbersome and inconsistent.
- Solution: Berzelius developed the system of using letters (usually the first one or two letters of the element’s Latin name) to represent elements (e.g., O for oxygen, Fe for iron, Au for gold). He also introduced using subscripts to indicate the number of atoms of each element in a compound (e.g., H₂O for water).
- Impact: This system, with minor modifications, is still used today. It provided a clear, concise, and universally understandable way to represent chemical formulas and equations.
- Accurate Determination of Atomic Weights (1807-1818):
- Problem: Dalton’s atomic weights were often inaccurate due to limitations in experimental techniques and assumptions.
- Solution: Berzelius devoted years to meticulously determining the atomic weights of many elements using precise analytical methods. He developed new techniques for analyzing chemical compounds and improved the accuracy of existing methods.
- Impact: His accurate atomic weights provided essential data for the development of chemical theory and for quantitative calculations in chemistry. They were crucial for validating (and refining) Dalton’s atomic theory.
- Discovery and Isolation of Elements:
- Discovered: Cerium (1803, independently with Wilhelm Hisinger), Selenium (1817), Thorium (1828).
- Isolated: Silicon, Zirconium, and Titanium (though not always in pure form).
- Impact: Expanded the known list of elements and contributed to understanding their properties.
- Electrochemical Dualism (1810s-1820s):
- Theory: Proposed that chemical compounds are formed by the combination of electrically positive and negative elements, held together by electrostatic attraction.
- Impact: This was an early attempt to explain chemical bonding based on electrical forces. While the theory was later superseded, it was essential to understanding chemical bonding.
- Isomerism and Catalysis (1830s):
- Isomerism (1830): Coined the term “isomerism” to describe compounds with the same chemical formula but different arrangements of atoms and different properties.
- Catalysis (1835): Coined the term “catalysis” to describe the phenomenon where a substance (a catalyst) speeds up a chemical reaction without being consumed itself.
- Impact: These concepts are fundamental to modern organic chemistry and industrial chemistry.
- Allotropy: He coined this word.
- Polymer: He coined this word.
- Protein Proposed name.
- Textbooks and Influence:
- ** Lärbok i Kemien (Textbook of Chemistry):** Wrote a highly influential textbook that went through many editions and was translated into several languages. This helped to disseminate his ideas and standardize chemical knowledge.
- Annual Reports: Published annual reports on the progress of chemistry, which were widely read and translated.
- Extensive Correspondence: Corresponded with leading chemists throughout Europe, influencing the development of the field.
- Laboratory: One of the best.
- Later Life and Legacy:
- Dominant Figure in Chemistry: Berzelius became one of the most dominant and respected figures in chemistry during the first half of the 19th century. He was known for his meticulous experimental work, theoretical insights, and organizational efforts.
- “Dictator of Chemistry”: Due to his influence and sometimes dogmatic views, he was sometimes called the “dictator of chemistry.”
- Honors and Awards: He received numerous honors and awards for his contributions to science.
- Death (1848): Died in Stockholm in 1848.
In summary, Jöns Jacob Berzelius was a giant of 19th-century chemistry. He systematized chemical notation, determined accurate atomic weights, discovered new elements, introduced fundamental concepts like isomerism and catalysis, and wrote influential textbooks. His work helped to establish chemistry as a modern, quantitative science and laid the foundation for many subsequent developments. He was a meticulous experimentalist, a brilliant theorist, and a powerful organizer, leaving a lasting legacy on the field. He brought order and precision to chemistry.
Jöns Jacob Berzelius, Standardization and Systematization, and Legacy. Table

Statue of Berzelius in the center of Berzelii Park, Stockholm
(Wiki Image By Jim.henderson – Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=81469213)
Let’s present Jöns Jacob Berzelius’s (1779-1848) contributions to chemistry in a structured table format, focusing on his role in standardization and systematization. This will clearly outline his key innovations and their lasting impact.
| Area of Contribution | Berzelius’s Key Actions and Innovations | Significance and Impact |
| Chemical Notation | – Developed the modern system of chemical notation: <br> – Used letters (usually the first one or two letters of the element’s Latin name) to represent elements (e.g., O for oxygen, Fe for iron, Au for gold). <br> – Used subscripts to indicate the number of atoms of each element in a compound (e.g., H₂O for water). <br> – Used + and – to represent what he viewed as the charge. | – Revolutionized chemical communication. <br> – Replaced the cumbersome and inconsistent symbols and abbreviations used previously. <br> – Provided a clear, concise, and universally understandable way to represent chemical formulas and equations. <br> – The foundation of the chemical notation system is still used today. |
| Atomic Weights | – Dedicated years to meticulously determining the atomic weights of many elements. <br> – Developed new and improved analytical techniques. <br> – Published several editions of his table of atomic weights, continually refining the values. | – Provided essential quantitative data for chemical calculations and for testing chemical theories (including Dalton’s atomic theory). <br> – His values were remarkably accurate for his time and far superior to Dalton’s initial estimates. <br> – Established the importance of precise measurement in chemistry. |
| Discovery of Elements | – Discovered cerium (1803, independently with Wilhelm Hisinger), selenium (1817), and thorium (1828). <br> – Isolated (or helped to isolate) silicon, zirconium, and titanium (though not always in perfectly pure form). | – Expanded the known list of elements. <br> – Contributed to understanding these new elements’ properties. |
| Chemical Theory | – Electrochemical Dualism: Proposed an early electrochemical theory of chemical bonding, suggesting that compounds are formed by the attraction of electrically positive and negative elements. | – An important early attempt to explain chemical bonding based on electrical forces. <br> – While the theory was later superseded, it was influential in its time and stimulated further research. |
| New Terminology | – Coined the term “isomerism” (1830): To describe compounds with the same chemical formula but different arrangements of atoms and different properties. <br> – Coined the term “catalysis” (1835): To describe the phenomenon where a substance (a catalyst) speeds up a chemical reaction without being consumed itself. <br> – Coined the terms “polymer” and “allotropy.” | – Introduced fundamental concepts into chemistry. <br> – Provided the vocabulary to describe and understand these important phenomena. <br> – Isomerism is crucial in organic chemistry. <br> – Catalysis is essential to many industrial processes and biological reactions. |
| Dissemination of Knowledge | – Lärbok i Kemien (Textbook of Chemistry): Wrote a highly influential textbook that went through many editions and was translated into several languages. <br> – Annual Reports: Published annual reports on the progress of chemistry, summarizing discoveries and developments. <br> – Extensive Correspondence: Maintained extensive correspondence with leading European chemists. | – Standardized chemical knowledge and terminology. <br> – Disseminated his discoveries and theories, as well as the work of other chemists. <br> – Became a central figure in the international chemical community, shaping the development of the field. <br> – His textbook was a major influence on chemical education. |
| Laboratory Practice | – Developed/improved many lab techniques. Introduced: filter paper, rubber tubing. | |
| Legacy | – “Father of Swedish Chemistry”: Considered the founder of modern chemistry in Sweden. <br> – One of the Founders of Modern Chemistry: His work on chemical notation, atomic weights, and new concepts had a profound and lasting impact on the field. <br> – Systematization and Quantification: He brought order, precision, and a quantitative approach to chemistry. <br> – Influence on Other Chemists: His work influenced generations of chemists, including his students and those who read his publications. | – His contributions were essential for transforming chemistry into a modern, quantitative science. <br> – His system of chemical notation is still used today. <br> – His concepts of isomerism and catalysis remain fundamental to chemistry. <br> – He was a towering figure in 19th-century chemistry. |
Export to Sheets
Key Takeaways:
- Systematizer: Berzelius’s most significant contribution was his systematization of chemistry. He brought order and clarity to a field characterized by inconsistent terminology and imprecise measurements.
- Experimentalist: He was a highly skilled and meticulous experimentalist who developed new techniques and made precise measurements.
- Communicator: He was a prolific writer and communicator, disseminating his ideas and others’ work through his textbooks, annual reports, and correspondence.
- Lasting Impact: His work on chemical notation, atomic weights, and new concepts laid the foundation for future chemical progress.
This table provides a structured and detailed overview of Jöns Jacob Berzelius’s contributions to chemistry, emphasizing his role in standardizing and systematizing the field. It highlights his key innovations, their impact, and his lasting legacy as one of the founders of modern chemistry. The format allows for a clear understanding of the breadth and depth of his work.
Berzelius Chemical Notation: Developed the modern system of chemical notation. Table
Jöns Jacob Berzelius’s development of the modern system of chemical notation was a monumental contribution to chemistry. It brought clarity and consistency to a field that was previously quite chaotic. Here’s a breakdown of his system and its significance:
Berzelius’s Chemical Notation
| Feature | Description | Significance |
| Elemental Symbols | * Used the initial letter(s) of the Latin names of elements. <br> * If multiple elements had the same initial letter, a second letter was added. <br> * Example: O for oxygen, Ca for calcium. | * Provided a concise and universal way to represent elements. <br> * Greatly simplified the writing of chemical formulas. <br> * His basic methodology is what is still used today. |
| Formula Composition | * Indicated the proportions of elements in a compound using numerical subscripts (though he originally used superscripts). <br> * Example: H₂O for water. | * Allows the precise representation of chemical compounds. <br> * Facilitated quantitative calculations in chemistry. <br> * Gave very clear insight into chemical compounds. |
| Organization of information | Berzelius’s notation allowed chemists to communicate chemical compositions accurately in written work. | The ability to easily communicate about chemical compounds greatly increased the advancement rate within chemistry science. |
Export to Sheets
Key Points:
- Standardization:
- Berzelius’s system brought much-needed standardization to chemical notation.
- Clarity and Precision:
- His symbols and formulas made it much easier to represent and communicate chemical information.
- Foundation for Modern Chemistry:
- His work laid the foundation for the chemical notation that is still used today.
- Improved Accuracy:
- Having a more standardized notation reduced errors in the recording of experimental data.
It is important to understand that while the placement of the numbers has changed from superscripts to subscripts, the core concept that Berzelius created is still the base of how we represent chemical formulas.
Berzelius Atomic Weights: Discovered cerium (independently with Wilhelm Hisinger), selenium (1817), and thorium, and isolated (or helped to isolate) silicon, zirconium, and titanium (though not always in perfectly pure form). Table
Let’s present Jöns Jacob Berzelius’s contributions related to atomic weights and the discovery/isolation of elements in a clear table format. This will separate these two areas of his work and highlight their interconnectedness.
Part 1: Atomic Weights
| Action | Details | Significance |
| Meticulous Determination of Atomic Weights | – Berzelius devoted over a decade (roughly 1807-1818, with later revisions) to systematically and painstakingly determining the atomic weights of a large number of elements. <br> – He used and improved existing analytical techniques and developed new ones. <br> – He emphasized precise measurements and repeated experiments to ensure accuracy. <br> – He expressed his results relative to oxygen (oxygen = 100), a different standard than Dalton’s (hydrogen = 1). | – Provided essential quantitative data for the development of chemical theory. Accurate atomic weights were crucial for: <br> – Verifying (and refining) Dalton’s atomic theory. <br> – Determining the correct formulas of compounds. <br> – Performing stoichiometric calculations (calculating the amounts of reactants and products in chemical reactions). <br> – His values were far more accurate than Dalton’s initial estimates due to Berzelius’s superior experimental skills and techniques. <br> – Established a standard for chemical analysis. |
| Publication of Atomic Weight Tables | – Published several editions of his table of atomic weights, constantly refining the values as he improved his techniques and obtained new data. | – Disseminated his results to the wider scientific community. <br> – His tables became a standard reference for chemists throughout Europe. |
| Impact of Oxygen Standard | Initially, I used hydrogen at 1, then oxygen at 100. | Eventually, it switched to oxygen = 16. |
Export to Sheets
Part 2: Discovery and Isolation of Elements
| Element | Year of Discovery/Isolation | Berzelius’s Role | Significance |
| Cerium | 1803 | – Independently co-discovered cerium with Wilhelm Hisinger (and, simultaneously, Martin Heinrich Klaproth). <br> – Analyzed the mineral cerite and identified a new element within it. | – Added a new element to the growing list of known elements. <br> – Demonstrated the power of chemical analysis to identify new substances. |
| Selenium | 1817 | – Discovered selenium while investigating impurities in sulfuric acid produced at a factory. | – Another new element added to the periodic table. <br> – Showed his keen observation skills and his ability to identify new substances even in unexpected places. |
| Thorium | 1828 | – Discovered thorium in a mineral sample from Norway. | – A further addition to the list of elements. <br> – Thorium later became important in the study of radioactivity (though this was long after Berzelius’s discovery). |
| Silicon | 1824 | – Isolated silicon in a relatively pure form (though not perfectly pure) by reducing potassium fluorosilicate with potassium. | – Silicon is a very abundant and important element (e.g., in semiconductors). |
| Zirconium | 1824 | – Isolated zirconium (in an impure form) by heating potassium heptafluorozirconate with potassium. | – Prepared the element. |
| Titanium | 1825 | Prepared somewhat pure metallic titanium. | – Although already discovered, prepared a pure form. |
Export to Sheets
Key Connections:
- Atomic Weights and New Elements: Berzelius’s work on determining accurate atomic weights was closely linked to his discovery and isolation of new elements. He needed to determine the atomic weights of these new elements to fit them into the developing understanding of chemical relationships.
- Analytical Chemistry: His discoveries directly resulted from his expertise in analytical chemistry – the art of separating and identifying the components of substances. He was a master of chemical analysis.
- Systematization: His work on atomic weights and new elements contributed to the overall systematization of chemical knowledge, laying the groundwork for Mendeleev’s later work on the periodic table.
This set of tables separates Berzelius’s work on atomic weights from his discovery and isolation of elements while also highlighting the crucial connections between these two areas of his research. It demonstrates how his meticulous experimental work, analytical skills, and drive to systematize chemical knowledge made him one of the most important figures in the history of chemistry.
Berzelius’s Discovery of Elements: Dedicated years to meticulously determining the atomic weights of many elements. Table
Jöns Jacob Berzelius’s dedication to determining accurate atomic weights was a cornerstone of his contributions to chemistry. His meticulous work provided a much-needed quantitative foundation for the field. Here’s a table summarizing his efforts:
Berzelius: Atomic Weight Determination
| Aspect | Description | Significance |
| Meticulous Measurements | * Berzelius dedicated years to performing highly precise experiments to determine the atomic weights of numerous elements. <br> * He placed great emphasis on the purity of his samples and the accuracy of his measurements. | * His work provided a reliable and extensive set of atomic weights, which were crucial for the advancement of stoichiometry and chemical calculations. <br> * His dedication set a standard for precision in chemical experimentation. |
| Extensive Data Collection | * He analyzed a vast number of compounds to obtain accurate data. <br> * His work was far more comprehensive than previous attempts to determine atomic weights. | * He significantly expanded the knowledge of atomic weights, providing a valuable resource for chemists. <br> * His data helped solidify the validity of Dalton’s atomic theory by providing experimental support. |
| Refining atomic weight knowledge | Berzelius’s work helped refine and correct earlier atomic weight measurements, leading to much more accurate data. | Accurate atomic weights are crucial for chemistry calculations, and his work’s accuracy greatly increased chemists’ ability to perform calculations. |
| Impact on Chemical Formulas | Having more accurate atomic weights allowed chemical formulas to become more accurate. | Because compounds comprise elements, accurate atomic weights allow for accurate compound formulas. |
Export to Sheets
Key Points:
- Quantitative Foundation:
- Berzelius’s work provided the quantitative foundation necessary for the development of modern chemistry.
- Accuracy and Precision:
- His emphasis on accuracy and precision set a new standard for chemical experimentation.
- Advancement of Stoichiometry:
- His atomic weight data was essential for the advancement of stoichiometry and chemical calculations.
It is clear that his drive for accuracy greatly impacted the progress of chemistry as a whole.
Berzelius Chemical Theory: Electrochemical Dualism. Table
Let’s present Jöns Jacob Berzelius’s electrochemical dualism theory of chemical bonding in a structured table format. This will outline the context, the key postulates of the theory, its strengths and limitations, and its eventual fate.
| Aspect | Details |
| Context (Pre-Berzelius) | – Early Ideas about Chemical Affinity: Chemists had long sought to understand what held atoms together in compounds (chemical affinity). Various theories existed, but none were fully satisfactory. <br> – Influence of Electrochemistry: The invention of the voltaic pile (battery) by Alessandro Volta in 1800 and early experiments on electrolysis (using electricity to decompose compounds) suggested a link between electricity and chemical bonding. Humphry Davy’s work was particularly influential. |
| Berzelius’s Motivations | – Explain Chemical Combination: To develop a comprehensive theory that could explain why elements combine in specific proportions to form compounds. <br> – Incorporate Electrical Phenomena: To integrate the new discoveries in electrochemistry into chemical theory. <br> – Systematize Chemistry: To provide a unifying framework for understanding chemical reactions. |
| Key Postulates of Electrochemical Dualism | – All atoms possess both positive and negative electrical charges. However, one type of charge is predominant in each element. <br> – Elements are classified as either electropositive (predominantly positive charge) or electronegative (predominantly negative charge). Oxygen was considered the most electronegative element. <br> – Chemical combination occurs due to the attraction between oppositely charged atoms. Electropositive elements combine with electronegative elements. <br> – The strength of the bond depends on the difference in electronegativity between the combining elements. <br> – Compounds can also have a residual electrical polarity, leading to further combinations (forming more complex compounds). |
| Examples | – Oxygen (strongly electronegative) combines with metals (electropositive) to form oxides. <br> – Sulfur (electronegative) combines with metals to form sulfides. <br> – Oxides can then combine with each other, based on their residual polarities (e.g., a basic oxide like Na₂O combining with an acidic oxide like SO₃ to form a salt, Na₂SO₄). |
| Strengths of the Theory (at the time) | – Explained Many Observations: It could explain many known chemical reactions, particularly the formation of inorganic compounds (acids, bases, salts). <br> – Provided a System: It provided a systematic way to classify elements and compounds based on their electrical character. <br> – Linked Chemistry and Electricity: It connected chemistry to the exciting new field of electricity. <br> – Influenced Nomenclature: It influenced Berzelius’s system of chemical nomenclature (e.g., the use of “-ide,” “-ite,” and “-ate” suffixes). |
| Limitations and Problems | – Difficulty with Organic Compounds: The theory struggled to explain the bonding in organic compounds (compounds containing carbon), which often involve combinations of elements with similar electronegativities. <br> – No Clear Explanation of How Charges Arise: The theory didn’t explain why atoms had these electrical charges or how they were distributed within the atom. <br> – Dualistic Nature: The strictly dualistic view (positive vs. negative) was too simplistic to account for all types of chemical bonding. <br> – Dumas’s Substitution Reactions: Discoveries in organic chemistry, particularly substitution reactions (where chlorine could replace hydrogen in organic compounds, seemingly contradicting the electronegativity differences), challenged the theory. |
| Eventual Fate | – Superseded by Later Theories: Electrochemical dualism was gradually superseded by more sophisticated theories of chemical bonding, particularly valence theory and, later, quantum mechanical explanations of bonding (covalent bonding, developed by G.N. Lewis and Linus Pauling). |
| Legacy | – Important Step in Understanding Bonding: While ultimately incorrect, electrochemical dualism was an important step in the development of chemical bonding theory. It was the first major attempt to explain bonding in terms of electrical forces. <br> – Influence on Nomenclature: His ideas influenced chemical nomenclature, and some aspects of his system are still reflected in modern naming conventions. <br> – Stimulated Research: The theory and the debates surrounding it stimulated further research into the nature of chemical bonding. |
Export to Sheets
Key Takeaways:
- First Major Electrical Theory of Bonding: Berzelius’s electrochemical dualism was the first comprehensive attempt to explain chemical bonding in terms of electrical forces.
- Success with Inorganic Compounds: It was relatively successful in explaining the formation of many inorganic compounds.
- Failure with Organic Compounds: It failed to adequately explain the bonding in organic compounds, which ultimately led to its downfall.
- Transitional Theory: It was a transitional theory that bridged the gap between earlier, less scientific ideas about chemical affinity and the later, more sophisticated theories of the 20th century.
This table provides a clear and structured overview of Berzelius’s electrochemical dualism, key principles, strengths and weaknesses, and ultimate fate. It highlights its historical importance as an early attempt to systematically explain chemical bonding, even though the theory was eventually superseded. The format facilitates understanding of this complex and now obsolete but historically significant chemical theory.
Berzelius New Terminology: Coined the term “isomerism,” “catalysis,” “polymer,” and “allotropy.” Table
Let’s create a table focusing on Jöns Jacob Berzelius’s contributions to chemical terminology, specifically his coining of the terms “isomerism,” “catalysis,” “polymer,” and “allotropy,” explaining their original context, definitions, and modern understanding.
| Term | Year Coined | Berzelius’s Original Definition/Context | Modern Definition/Understanding | Significance |
| Isomerism | 1830 | Berzelius observed that some compounds had the same chemical formula (same number and types of atoms) but different properties. He coined “isomerism” (from Greek isos = equal, meros = part) to describe this phenomenon. He initially observed this with tartaric acid and racemic acid. | Compounds with the same molecular formula but different structural formulas (different arrangements of atoms) and, consequently, different physical and/or chemical properties. We now recognize many different types of isomerism (structural isomers, stereoisomers, etc.). | – Introduced a fundamental concept in organic chemistry (and later inorganic chemistry). <br> – Showed that chemical formula alone is not enough to define a compound; the arrangement of atoms is crucial. <br> – Led to a deeper understanding of molecular structure. |
| Catalysis | 1835 | Berzelius observed that some substances could speed up chemical reactions without being consumed themselves in the process. He coined “catalysis” (from Greek kata = down, lyein = loosen) to describe this phenomenon, drawing an analogy to the decomposition of substances. He saw it as a force. | A substance that increases the rate of a chemical reaction without itself undergoing any permanent chemical change. Catalysts work by providing an alternative reaction pathway with a lower activation energy. We now understand catalysis in terms of chemical kinetics and reaction mechanisms. | – Introduced a fundamental concept in chemistry. <br> – Recognized the importance of catalysts in many chemical processes, both in the lab and in industry. <br> – Catalysis is essential for many industrial chemical processes and for biological reactions (enzymes are biological catalysts). |
| Polymer | 1833 | Berzelius used “polymer” to refer to compounds with the same empirical formula but different molecular weights. He considered benzene to be a polymer of acetylene. | A substance composed of macromolecules, large molecules made up of repeating structural units (monomers) connected by chemical bonds. Modern understanding includes a vast array of natural and synthetic polymers with diverse structures and properties. | The term has undergone a major change. |
| Allotropy | ~1840 | He used this to mean different forms of an element. | Different structural forms of the same element in the same physical state (e.g., diamond and graphite are allotropes of carbon; oxygen gas (O₂) and ozone (O₃) are allotropes of oxygen). | The term has survived. |
| Protein | 1838 | Suggested the name to Mulder. | The term is still used. |
Export to Sheets
Key Points:
- Berzelius’s Insight: Berzelius was a keen observer of chemical phenomena and recognized the need for new terminology to describe them accurately.
- Lasting Impact: The terms “isomerism” and “catalysis” are still used today, with their meanings largely unchanged (though our understanding of the underlying mechanisms has deepened considerably). “Allotropy” is also still a standard term. “Polymer,” while still used, has changed in meaning.
- Conceptual Advances: These terms weren’t just labels; they represented new concepts that helped to organize and understand chemical phenomena.
- Organic Focus Isomerism became key to organic chemistry.
This table presents Berzelius’s key contributions to chemical terminology, showing how he introduced new terms to describe critical phenomena he observed. It highlights both his original definitions and the modern understanding of these terms, demonstrating the lasting impact of his work on the language and concepts of chemistry. The format allows for a direct comparison between his original ideas and our current understanding.
Dmitri Mendeleev (1834-1907) – Periodic Law and Table

Dmitri Ivanovich Mendeleev
(Wiki ImageBy Ivan Kramskoi – Музей-архив Д. И. Менделеева (СПбГУ), 2009, Public Domain, https://commons.wikimedia.org/w/index.php?curid=7507431)
Mendeleev Quotes Table
Dmitri Mendeleev’s work was driven by a deep desire to find order in the seemingly chaotic world of elements. His quotes often reflect this drive, as well as his dedication to the scientific method. Here’s a table of some of his notable quotes:
Dmitri Mendeleev Quotes
| Quote | Theme/Context |
| “The periodic law discovered by me, being not merely a summary of facts, but a reliable and fruitful method of investigation, may serve as a means of further progress in the discovery of new elements.” | Periodic law, scientific discovery, and the predictive power of science. |
| “There can be no scientific truth without experiment. There can be no science without control.” | The scientific method, the importance of experimentation, and control. |
| “The edifice of science not only requires material, but also a plan. Without the material, the plan alone is but a castle in the air—a mere possibility; whilst the material without a plan is but useless matter.” | Science is a process that needs structure, data, and the balance of theory and experiment. |
| “To discover a law of nature is a far higher achievement than to invent a machine.” | The value of scientific discovery, laws of nature vs. inventions. |
| “I saw in a dream, where all the elements fell into place as required. Only once did I see a correction needed. The elements arranged themselves.” | This is a more anecdotal quote, but it shows Mendeleev’s dedication to his work. |
| “Work, look for peace and calm in work: you will find it nowhere else.” | On the value of dedicated work. |
| “In science, we must all submit not to what seems to us attractive from one point of view or another, but to what represents an agreement between theory and experiment.” | The Need for Objective Thinking in Science. |
Export to Sheets
Key Observations:
- Mendeleev’s quotes emphasize the importance of the periodic law as a tool for discovery.
- He stressed the necessity of rigorous experimentation and the scientific method.
- His statements reveal his deep belief in the underlying order of the natural world.
- He shows that hard work and dedication are extremely important for scientific discovery.
Mendeleev YouTube Video
- The genius of Mendeleev’s periodic table – Lou Serico by TED-Ed: 3,272,234 views (http://www.youtube.com/watch?v=fPnwBITSmgU)
- Mendeleev’s Periodic Table by SciShow: 542,419 views (http://www.youtube.com/watch?v=-wu0LixSBpk)
- Mendeleev Periodic Table by Manocha Academy: 559,067 views (http://www.youtube.com/watch?v=BemuunA0Fzc)
- Dmitri Mendeleev: Great Minds by SciShow: 515,787 views (http://www.youtube.com/watch?v=IgA37CNa7Ow)
- Mendeleev and the Periodic Table | Properties of Matter | Chemistry | FuseSchool by FuseSchool – Global Education: 80,041 views (http://www.youtube.com/watch?v=L4M2VKASI1Q)
Dmitri Mendeleev History

Sculpture in honor of Mendeleev and the periodic table, located in Bratislava, Slovakia
(Wiki Image By https://www.flickr.com/people/mmmdirt/ – https://www.flickr.com/photos/mmmdirt/279349599, CC BY-SA 2.0, https://commons.wikimedia.org/w/index.php?curid=1526889)
Dmitri Ivanovich Mendeleev (1834-1907) was a Russian chemist and inventor best known for formulating the periodic law and creating his version of the periodic table of elements. His work was a monumental achievement in chemistry, providing a fundamental organizing principle for understanding the elements and their properties.
- Early Life and Education (1834-1859):
- Born in Tobolsk, Siberia (1834): The youngest of a large family (accounts vary on the exact number of siblings, ranging from 11 to 17). His father was a teacher.
- Early Hardships: His father died when Dmitri was young, and the family faced financial difficulties. His mother played a crucial role in supporting his education.
- Education in St. Petersburg: His mother recognized his intellectual potential and, with considerable effort, got him enrolled in the Main Pedagogical Institute in St. Petersburg (the capital of Russia at the time).
- University Studies: He excelled in science, particularly chemistry, at the Pedagogical Institute and St. Petersburg University.
- Early Research: His early research focused on organic chemistry.
- Studies Abroad (1859-1861): He spent two years (1859-1861) studying in Heidelberg, Germany, under renowned chemists Robert Bunsen and Gustav Kirchhoff. This exposed him to the latest developments in European chemistry. He worked on the capillarity and surface tension of liquids. Importantly, he attended the Karlsruhe Congress (1860).
- The Karlsruhe Congress (1860) and its Influence:
- International Chemical Congress: Mendeleev attended the Karlsruhe Congress in 1860, the first international chemistry conference. This was a pivotal event in his career.
- Clarification of Atomic Weights: The congress addressed the confusion surrounding atomic weights, molecular weights, and chemical formulas. Stanislao Cannizzaro presented a clear distinction between atomic and molecular weights based on Avogadro’s hypothesis (which had been largely ignored for decades).
- Impact on Mendeleev: Cannizzaro’s ideas had a profound impact on Mendeleev. Clarifying atomic weights provided the crucial data he needed to develop his periodic system.
- Development of the Periodic Table (1869):
- Organizing the Elements: Mendeleev began working on organizing the known elements systematically. He was writing a textbook and looking for a logical way to present the elements.
- Arrangement by Atomic Weight: He initially arranged the elements in order of increasing atomic weight (as others had done before him).
- Periodicity: He noticed that elements with similar chemical properties appeared regularly (periodicity). This was his key insight.
- The Periodic Table: He created a table where elements with similar properties were grouped. He boldly left gaps in his table for undiscovered elements.
- Predictions: Mendeleev used his periodic table to predict the existence and properties of several undiscovered elements (which he called eka-aluminum, eka-boron, and eka-silicon – “eka” meaning “one beyond” in Sanskrit).
- Publication (1869): In 1869, he published his periodic table and the periodic law in a Russian journal and later in a German journal (Zeitschrift für Chemie, 1869), bringing them to the attention of the wider scientific community.
- Confirmation and Acceptance (1870s-1880s):
- Discovery of Predicted Elements: The discovery of gallium (1875), scandium (1879), and germanium (1886), with properties that closely matched Mendeleev’s predictions, provided stunning confirmation of his periodic law and his table.
- Gradual Acceptance: The periodic table gradually gained acceptance among chemists, becoming recognized as a fundamental organizing principle of chemistry.
- Revisions and Refinements: The periodic table was revised and refined over time, particularly with the discovery of new elements and the understanding of atomic structure (atomic number, not atomic weight, is the true basis for the periodic law). However, the basic structure and principles remained the same.
- Later Career and Legacy:
- Professor at St. Petersburg University: Mendeleev taught chemistry at St. Petersburg University for many years, influencing generations of Russian chemists.
- Extensive Research: He continued researching various topics, including chemical solutions, petroleum, and the expansion of liquids with heat.
- Resignation (1890): In 1890, he resigned from the university to protest the government’s repressive policies toward students.
- Director of the Bureau of Weights and Measures (1893): Appointed director of the Bureau of Weights and Measures, where he worked on standardizing measurement units.
- Nobel Prize Nomination: He was nominated for the Nobel Prize in Chemistry but narrowly missed receiving it (reportedly due to the influence of Svante Arrhenius, who held a grudge against Mendeleev). The Nobel Committee considers this a significant oversight.
- Death (1907): Died in St. Petersburg in 1907.
- Element 101: Named after him.
- Significance and Impact:
- Fundamental Organizing Principle: The periodic table is one of chemistry’s most important organizing principles. It provides a framework for understanding the relationships between elements and their properties.
- Predictive Power: Mendeleev’s periodic table was not just a classification system; it had predictive power, allowing him to anticipate the existence and properties of undiscovered elements.
- Foundation for Future Discoveries: The periodic table provided a roadmap for future research, guiding the discovery of new elements and understanding their properties.
- Lasting Legacy: The periodic table remains a cornerstone of chemical education and research, a testament to Mendeleev’s genius and insight. It is one of the most recognizable and iconic images in science.
Dmitri Mendeleev’s creation of the periodic table was a landmark achievement in the history of chemistry. He transformed the field from a collection of disparate facts into a coherent and organized system based on a fundamental underlying principle. His work continues to shape our understanding of the elements and their interactions, and his legacy as one of the most outstanding chemists of all time is secure.
Dmitri Mendeleev, Periodic Law and Table, and Legacy. Table
Mendeleev’s 1871 periodic table
(Wiki Image By The original uploader was Den fjättrade ankan at Swedish Wikipedia. – Källa:Dmitrij Ivanovitj Mendelejev (1834 – 1907). Originally from sv.wikipedia; description page is/was here., Public Domain, https://commons.wikimedia.org/w/index.php?curid=1574857)
Let’s present Dmitri Mendeleev’s story, his development of the periodic law and table, and his legacy in a structured table format. This will delineate the context, his actions, the key features of his system, and the long-term impact.
| Aspect | Details |
| Context (Pre-Mendeleev) | – Many Known Elements: By the mid-19th century, over 60 elements were known, with diverse properties. <br> – Atomic Weights: Chemists had determined the relative atomic weights of many elements. <br> – Previous Attempts at Classification: Several chemists had attempted to classify the elements based on their properties (e.g., Döbereiner’s triads, Newlands’ law of octaves), but with limited success. <br> – Confusion and Lack of System: There was no universally accepted system for organizing the elements, and their relationships were poorly understood. |
| Mendeleev’s Background | – Russian Chemist (1834-1907): Professor of chemistry at St. Petersburg University. <br> – Textbook Author: Writing a textbook, Principles of Chemistry, and needed a logical way to organize the elements. <br> – Karlsruhe Congress (1860): Attended the Karlsruhe Congress, which clarified the concepts of atomic and molecular weights (influenced by Cannizzaro’s work). This provided crucial data. |
| Development of the Periodic Law and Table (1869) | – Arrangement by Atomic Weight: Mendeleev initially arranged the elements in order of increasing atomic weight. <br> – Observation of Periodicity: He noticed that elements with similar chemical properties appeared at regular intervals (periodicity). <br> – Creation of the Table: He organized the elements into a table, grouping elements with similar properties in the same vertical columns (groups) and arranging them horizontally by increasing atomic weight. <br> – Leaving Gaps: He boldly left gaps in his table for undiscovered elements, believing that elements with properties to fill those gaps must exist. <br> – Predictions: He used his table to predict the existence and properties (atomic weight, density, chemical behavior) of several undiscovered elements (eka-aluminum, eka-boron, and eka-silicon). <br> – Periodic Law: He formulated the periodic law: “The properties of the elements are periodic functions of their atomic weights.” (Later refined to atomic number). <br> – Publication (1869): Published his table and law in Russian and German journals. |
| Key Features of Mendeleev’s Table | – Vertical Groups: Elements in the same vertical column (group) have similar chemical properties. <br> – Horizontal Periods: Elements in the same horizontal row (period) show a gradual property change. <br> – Gaps for Undiscovered Elements: His confidence in the periodic law was a key distinguishing feature. <br> – Predictions: Allowed for predictions about the properties of undiscovered elements. <br> – Flexibility: In a few cases, he deviated from strict atomic weight order to maintain the grouping of elements with similar properties (e.g., tellurium and iodine). The discovery of isotopes later explained this. |
| Confirmation and Acceptance | – Discovery of Gallium (1875), Scandium (1879), and Germanium (1886): These elements were discovered, and their properties closely matched Mendeleev’s predictions for eka-aluminum, eka-boron, and eka-silicon. <br> – Widespread Acceptance: His predictions’ confirmation led to the scientific community’s widespread acceptance of the periodic table and the periodic law. |
| Later Developments (Not by Mendeleev) | – Discovery of Noble Gases: The discovery of the noble gases (argon, helium, etc.) in the late 19th century added a new group. <br> – Atomic Number: The discovery of the atomic nucleus and the concept of atomic number (the number of protons in the nucleus) provided a more fundamental basis for the periodic law (Moseley’s work). <br> – Quantum Mechanics: The development of quantum mechanics in the 20th century explained the underlying reasons for the periodic trends in element properties based on electron configurations. |
| Mendeleev’s Legacy | – Fundamental Organizing Principle: The periodic table is one of chemistry’s most fundamental organizing principles. <br> – Predictive Tool: It remains a powerful tool for predicting the properties of elements and understanding chemical reactions. <br> – Cornerstone of Chemical Education: It is a worldwide chemical education and research cornerstone. <br> – Symbol of Scientific Insight: Mendeleev’s periodic table is a testament to the power of scientific reasoning and the ability to find order and patterns in the natural world. <br> – Element 101 (Mendelevium): Named in his honor. |
Export to Sheets
Key Takeaways:
- Periodicity: Mendeleev’s key insight was the periodicity of element properties.
- Bold Predictions: His willingness to leave gaps and predict the properties of undiscovered elements was crucial to the acceptance of his table.
- Confirmation: The discovery of the predicted elements provided strong evidence for the validity of the periodic law.
- Lasting Impact: The periodic table remains a fundamental tool for chemists and a testament to Mendeleev’s genius.
This table provides a comprehensive and structured overview of Dmitri Mendeleev’s life, his periodic law and table development, and his lasting legacy in chemistry. It highlights the key steps in his discovery, the features of his table, the confirmation of his predictions, and the ongoing importance of his work. The format allows for a clear understanding of the progression of his ideas and their profound impact on science.
Mendeleev Fundamental Organizing Principle: The periodic table is one of chemistry’s most fundamental organizing principles. Table
Dmitri Mendeleev’s creation of the periodic table stands as a monumental achievement in chemistry. Its power lies in its ability to organize a vast amount of chemical information into a coherent and predictive system. Here’s a table highlighting the key aspects of the periodic table as a fundamental organizing principle:
Mendeleev’s Periodic Table: A Fundamental Organizing Principle
| Aspect | Description | Significance |
| Organization by Periodic Properties | * Elements are arranged in rows (periods) and columns (groups) based on recurring similarities in their chemical properties. <br> * Mendeleev’s initial organization was primarily based on atomic weights, but it also heavily weighed chemical properties. <br> * The modern table is organized by atomic number. | * Provides a framework for understanding the relationships between elements. <br> * Reveals trends in properties such as electronegativity, ionization energy, and atomic radius. <br> * Simplifies the study of chemistry by grouping elements with similar behaviors. |
| Predictive Power | * Mendeleev left gaps in his table for undiscovered elements, predicting their properties with remarkable accuracy. <br> * The subsequent discovery of gallium, germanium, and scandium validated his predictions. | * Demonstrated the predictive power of scientific theories. <br> * Strengthened the acceptance of the periodic law as a fundamental principle. <br> * Showed that scientific principles can be used to make highly accurate predictions about the natural world. |
| Foundation for Modern Chemistry | * The periodic table is a cornerstone of chemical education and research. <br> * It provides a basis for understanding chemical bonding, reactivity, and the properties of materials. | * Serves as an indispensable tool for chemists, educators, and students. <br> * Guides the discovery of new elements and the development of new materials. <br> * Fosters a deeper understanding of the nature of matter. |
| Modern refinements | Mendeleev organized his table by atomic weight. Modern tables are organized by atomic number or number of protons. | Modern organizations more accurately reflect the patterns of chemical behavior. It helps to explain isotopes. |
Export to Sheets
Key Points:
- Order from Chaos:
- Mendeleev’s table brought order to the previously scattered knowledge of the elements.
- Predictive Success:
- The accuracy of his predictions was a powerful testament to the validity of his work.
- Lasting Impact:
- The periodic table remains an essential tool in chemistry today.
Mendeleev Predictive Tool: It remains a powerful tool for predicting the properties of elements and understanding chemical reactions. Table
You’re absolutely right. Mendeleev’s periodic table wasn’t just a static arrangement of elements; it’s a dynamic and predictive tool that continues to be invaluable. Here’s a table detailing its predictive power:
Mendeleev’s Periodic Table: A Predictive Tool
| Predictive Aspect | Description | Significance |
| Prediction of Undiscovered Elements | * Mendeleev left gaps in his table, indicating the existence of undiscovered elements. <br> * He predicted their properties, including atomic weights, densities, and chemical behaviors. | * The discovery of gallium, germanium, and scandium, with properties closely matching his predictions, validated the periodic law. <br> * It demonstrated the table’s power to guide scientific discovery. |
| Prediction of Chemical Properties | * Elements within the same group (column) exhibit similar chemical behaviors. <br> * The table helps predict how elements will react with each other and form compounds. | * Simplifies the study of chemical reactions by allowing chemists to infer properties based on an element’s position in the table. <br> * Aids in the design of chemical experiments and the development of new materials. |
| Prediction of Physical Properties | * Trends in physical properties like atomic radius, ionization energy, and electronegativity can be observed across periods and down groups. <br> * The table provides a visual representation of these trends. | * Allows scientists to estimate and understand the physical characteristics of elements. <br> * Essential for fields like materials science and solid-state physics. |
| Predicting new compounds | By examining the properties of nearby elements, chemists can make predictions about how undiscovered elements or combinations of elements may behave. | Speeds up research, saves resources, and increases the speed of scientific discoveries. |
Export to Sheets
Key Points:
- Validation of the Periodic Law:
- The successful prediction of undiscovered elements and their properties solidified the acceptance of the periodic law.
- Guidance for Chemical Research:
- The table serves as an indispensable tool for chemists in predicting chemical behavior and designing experiments.
- Foundation for Materials Science:
- The predictive power of the periodic table is vital in materials science for the development of new substances with desired properties.
- Continuing Relevance:
- Even with the development of quantum mechanics, the periodic table continues to be a cornerstone of chemical thinking and prediction.
Mendeleev Cornerstone of Chemical Education: It is a worldwide cornerstone of chemical education and research. Table
You’re right. The periodic table isn’t just a tool for professional chemists; it’s a fundamental part of chemical education worldwide. Here’s a table summarizing its role as a cornerstone of learning and research:
Mendeleev’s Periodic Table: Cornerstone of Chemical Education and Research
| Aspect | Description | Significance |
| Foundation for Chemical Concepts | * The periodic table provides a framework for introducing fundamental chemical concepts like atomic structure, bonding, and reactivity. <br> * It’s often one of the first things students encounter in chemistry classes. | * Simplifies the teaching and learning of chemistry by logically organizing information. <br> * Allows students to see the connections between different elements and their properties. <br> * Allows a quick visual aid when working on chemical equations or attempting to predict the outcome of a reaction. |
| Tool for Learning and Discovery | * Students use the periodic table to explore trends in chemical properties and make predictions. <br> * Researchers use it to guide the discovery of new materials and chemical reactions. | * Encourages active learning and critical thinking. <br> * Fosters a sense of curiosity and discovery about the natural world. <br> * Aids in the research and development of new technologies and materials. |
| International Standard | * The periodic table is universally used in chemistry education and research around the globe. <br> * It provides a common language for chemists worldwide. | * Facilitates communication and collaboration among scientists from different countries. <br> * Ensures consistency in chemical terminology and understanding. <br> * Promotes international scientific exchange. |
| Development of a deeper understanding | Students can use the periodic table to deepen their understanding of electron configurations, the nature of chemical bonds, and the forces that govern molecular interactions. | It increases fundamental understanding, which then allows for an increase in more advanced knowledge of chemical topics. |
Export to Sheets
Key Points:
- Universal Tool:
- The periodic table transcends cultural and linguistic barriers, serving as a universal language for chemists.
- Educational Foundation:
- It forms the bedrock upon which much of chemistry education is built.
- Research Guide:
- It remains an essential tool for cutting-edge chemical research.
- Continual Evolution: While the layout may not drastically change, our understanding of the elements and their relationships continues to deepen.
Mendeleev Symbol of Scientific Insight: Mendeleev’s periodic table is a testament to the power of scientific reasoning and the ability to find order and patterns in the natural world. Table
You’ve captured the essence of Mendeleev’s achievement perfectly. His periodic table is not just a scientific tool; it’s a symbol of the human capacity for insightful observation and logical deduction. Here’s a table that highlights this symbolic significance:
**Mendeleev’s Periodic Table: Symbol of Scientific Insight
| Aspect | Description | Significance |
| Discovery of Underlying Order | * Mendeleev’s table revealed the inherent order and patterns within the seemingly disparate elements. <br> * It demonstrated that the elements are not randomly scattered, but follow a coherent system. | * Showed that even complex natural phenomena can be understood through careful observation and logical analysis. <br> * Inspired generations of scientists to seek deeper patterns in the natural world. <br> * Acts as a wonderful example of the ability of the human mind to find previously undiscovered patterns. |
| Predictive Validation | * The successful prediction of undiscovered elements validated the power of scientific reasoning. <br> * It demonstrated that scientific theories can be used to make accurate predictions about the unknown. | * Strengthened the belief in the reliability of the scientific method. <br> * Emphasized the importance of testing theories through experimentation. <br> * Reinforces the idea that well-developed scientific models can predict phenomena yet to be observed. |
| Triumph of Logical Deduction | * Mendeleev’s work was a triumph of logical deduction based on existing data and careful consideration of chemical properties. <br> * It showed that scientific breakthroughs can be achieved through careful thought and analysis. | * Celebrates the power of the human mind to discern patterns and connections. <br> * Underscores the importance of critical thinking and analytical skills in scientific endeavors. <br> * A powerful example of how intellectual rigor can produce incredibly insightful results. |
| A lasting legacy | The continued use and refinement of the periodic table show the power of careful observation, the importance of predictive models, and the possibility of scientific ideas evolving over time. | It shows that even theories created long ago can be relevant in modern-day applications. |
Export to Sheets
Key Points:
- Human Ingenuity:
- The periodic table stands as a testament to human ingenuity and the pursuit of knowledge.
- The Power of Patterns:
- It highlights the importance of recognizing patterns in nature.
- Foundation for Discovery:
- It’s a reminder that fundamental discoveries can pave the way for future scientific progress.
Mendeleev Element 101 (Mendelevium): Named in his honor. Table
It’s a fitting tribute that Dmitri Mendeleev, the creator of the periodic table, has an element named in his honor. Here’s a table outlining the key facts about Mendelevium:
Mendelevium (Md) – Element 101
| Feature | Description | Significance |
| Atomic Number | 101 | Indicates the number of protons in the nucleus. |
| Symbol | Md | Abbreviation used to represent the element. |
| Discovery | 1955 | Discovered by Albert Ghiorso, Glenn T. Seaborg, Gregory Robert Choppin, Bernard G. Harvey, and Stanley G. Thompson at the University of California, Berkeley. |
| Naming | Named after Dmitri Mendeleev | Honoring the creator of the periodic table. |
| Category | Actinide, transuranium element | Placing it within the f-block of the periodic table. |
| Production | Synthetic | Produced in particle accelerators. |
| Properties | Radioactive | All isotopes are unstable. |
| Uses | Primarily research | Due to its instability and limited availability. |
| Key isotopes | 258Md | longest-lived isotope, with a half-life of 51.5 days. |
| The discovery method | bombarded einsteinium-253 with alpha particles | Producing Mendelevium 256. |
Export to Sheets
Key Points:
- Tribute to a Pioneer:
- The naming of Mendelevium acknowledges Dmitri Mendeleev’s profound impact on chemistry.
- Synthetic Element:
- Mendelevium is a synthetic element, meaning it’s not found naturally.
- Radioactive Nature:
- Like many heavy elements, Mendelevium is radioactive.
- Scientific Research:
- Because very small amounts have been made, its uses are limited to scientific research.
I hope that this information is helpful.
Marie (1867-1934) and Her Husband Pierre (1859 – 1906) Curie – Radioactivity

Pierre and Marie Skłodowska-Curie, 1895
(Wiki Image By Unknown author – [1], Public Domain, https://commons.wikimedia.org/w/index.php?curid=60656702)
Marie and Pierre Curie Quotes Table
Let’s present a selection of quotes from Marie and Pierre Curie and quotes about them in a table format. This will provide insights into their personalities, scientific work, dedication, and challenges. I’ll include context and significance for each quote.
| Quote Number | Quote | Source | Speaker (if applicable) | Context/Significance |
| 1 | “I am among those who think that science has great beauty.” | Widely attributed | Marie Curie | Expresses her passion for science and her aesthetic appreciation of its elegance and power. |
| 2 | “One never notices what has been done; one can only see what remains to be done…” | Widely attributed, often cited as reflecting her dedication to research | Marie Curie | Highlights her relentless pursuit of knowledge and her focus on the future of scientific inquiry. |
| 3 | “Be less curious about people and more curious about ideas.” | Widely attributed | Marie Curie | Reflects her focus on intellectual pursuits and her dedication to scientific discovery. |
| 4 | “Nothing in life is to be feared, it is only to be understood. Now is the time to understand more, so that we may fear less.” | Widely attributed | Marie Curie | Shows belief in the importance of reason. |
| 5 | “A scientist in his laboratory is not a mere technician: he is also a child confronting natural phenomena that impress him as though they were fairy tales.” | Widely attributed | Marie Curie | Shows how she approached her work. |
| 6 | “We must not forget that when radium was discovered, no one knew that it would prove useful in hospitals. The work was one of pure science.” | From a lecture at Vassar (1921) | Marie Curie | On the nature of science. |
| 7 | “Life is not easy for any of us. But what of that? We must have perseverance and above all confidence in ourselves. We must believe that we are gifted for something and that this thing must be attained.” | Marie Curie | Perseverance. | |
| 8 | “[On their working conditions] …we had no money, no laboratory and no help…and yet it was in this miserable old shed that we passed the best and happiest years of our lives…” | (Representative quote, reflecting their dedication despite hardship) | Both | Shows their love and dedication. |
| 9 | “Pierre Curie was a man of genius, a scientist of the first rank… But he was also a dreamer and an idealist, and he put his genius at the service of humanity.” | (Representative quote, reflecting assessments of Pierre Curie) | (Biographers and Historians) | Summarizes Pierre Curie’s scientific brilliance and his humanitarian values. |
| 10 | “Marie Curie is, of all celebrated beings, the only one whom fame has not corrupted.” | Albert Einstein – attributed | Albert Einstein | Shows respect for Marie Curie |
Export to Sheets
Key Takeaways:
- Passion for Science: The quotes reveal Marie Curie’s deep passion for science and her belief in its power and beauty.
- Dedication and Perseverance: They highlight her dedication to research, her perseverance in the face of adversity, and her focus on the long-term goals of science.
- Collaboration: The quotes emphasize the close collaboration between Marie and Pierre Curie, their shared dedication, and their complementary skills.
- Humility and Idealism: They reflect a sense of humility and a commitment to using science for the benefit of humanity.
- Overcoming Obstacles: Marie Curie’s life and work exemplify the challenges faced by women in science in the late 19th and early 20th centuries and her success in overcoming those obstacles.
This table provides a structured and informative presentation of key quotes from and about Marie and Pierre Curie, offering context and explanation for each. It allows for a deeper understanding of their personalities, their scientific values, and the significance of their work. The quotes, combined with the historical context, paint a picture of two extraordinary scientists who were driven by a passion for discovery and a commitment to advancing human knowledge.
Marie and Pierre Curie YouTube Video
- The genius of Marie Curie – Shohini Ghose by TED-Ed: 3,969,026 views (http://www.youtube.com/watch?v=w6JFRi0Qm_s)
- Marie Curie | Discovering Radioactivity | First Woman to Win the Nobel Prize by Little Fox – Kids Stories and Songs: 296,528 views (http://www.youtube.com/watch?v=1o35t1DfXCE)
- Marie and Pierre Curie by Sigma Documentaries: 16,638 views (http://www.youtube.com/watch?v=82Oj5qyY1F0)
- How madam marie curie and pierre curie discovered radioactivity?||ANIMATION||RADIOACTIVITY by budding scientist: 64,495 views (http://www.youtube.com/watch?v=w7RoFj3q4_U)
- How Power Couple Marie and Pierre Curie Revolutionized Science with Radium and Polonium by Histories and Biographies: 349 views (http://www.youtube.com/watch?v=zd6uJUEs9tc)
Marie and Pierre Curie History

Curie in a mobile X-ray vehicle, c. 1915
(Wiki Image By Unknown author – Eve Curie: Madame Curie. S. 329 [1], Public Domain, https://commons.wikimedia.org/w/index.php?curid=6266984)
Marie Curie (1867-1934) and Pierre Curie (1859-1906) were pioneering scientists whose collaborative work on radioactivity revolutionized physics and chemistry and profoundly impacted medicine and other fields. Their story is one of scientific partnership, dedication, and groundbreaking discovery, achieved despite significant challenges.
- Marie Curie (Before Meeting Pierre):
- Early Life and Education (1867-1891):
- Born Maria Skłodowska in Warsaw, Poland (then part of the Russian Empire).
- The family faced hardship under Russian rule. Her father was a teacher of mathematics and physics.
- Exceptional students faced barriers to higher education as women in Poland.
- She worked as a governess to support herself and her sister Bronisława’s medical studies in Paris.
- Secret education: Involved in the “Flying University”
- Move to Paris (1891):
- Moved to Paris to study physics and mathematics at the Sorbonne (University of Paris).
- Lived in poverty but was dedicated to her studies.
- Earned degrees in physics and mathematics.
- Pierre Curie (Before Meeting Marie):
- Early Life and Education (1859-1895):
- Born in Paris, France.
- He was educated at home by his father, a physician.
- Showed an early aptitude for science, particularly physics.
- Early Research:
- Worked at the School of Physics and Chemistry in Paris.
- Piezoelectricity (1880): Discovered piezoelectricity (with his brother Jacques Curie): generating an electric charge in certain crystals when subjected to mechanical stress. This discovery has critical applications in various devices.
- Magnetism: Studied the effects of temperature on magnetism (Curie’s Law).
- Collaboration and Discoveries (1895-1906):
- Marriage (1895): Marie and Pierre Curie married in 1895, forming a scientific partnership to change the world.
- Henri Becquerel’s Discovery (1896): Henri Becquerel discovered that uranium salts emitted rays that could penetrate opaque materials and fog photographic plates. This was the first observation of what would later be called radioactivity.
- Marie Curie’s Doctoral Research: For her doctoral research, Marie Curie chose to investigate Becquerel’s rays, a bold and unconventional choice at the time.
- Key Discoveries:
- Radioactivity: Marie Curie coined the term “radioactivity” to describe the phenomenon of spontaneous radiation emission from certain elements.
- Polonium and Radium (1898): Working with pitchblende (a uranium ore), Marie and Pierre Curie discovered two new radioactive elements: polonium (named after Marie’s native Poland) and radium. This was a painstaking and physically demanding process involving the processing of tons of pitchblende in poor laboratory conditions.
- Properties of Radioactive Elements: They studied the properties of these new elements, including their ability to ionize air and emit radiation.
- The realization that Radioactivity is an Atomic Property: Marie Curie hypothesized (correctly) that radioactivity was an atomic property, not a chemical property, challenging the prevailing view of the atom as indivisible.
- Nobel Prize in Physics (1903): Marie and Pierre Curie shared the Nobel Prize in Physics with Henri Becquerel for their work on radioactivity. Marie Curie was the first woman to win a Nobel Prize.
- Continued Work: Both continued to research the nature of these new elements.
- Pierre Curie’s Death and Marie Curie’s Continued Work (1906-1934):
- Pierre Curie’s Death (1906): Pierre Curie was tragically killed in a street accident in Paris, run over by a horse-drawn carriage.
- Marie Curie’s Grief and Determination: Despite her profound grief, Marie Curie was determined to continue their work.
- Succession to Pierre’s Professorship: She took over Pierre’s professorship at the Sorbonne, becoming the first woman to hold this position.
- Isolation of Pure Radium (1910): Marie Curie successfully isolated pure metallic radium, further confirming its elemental nature.
- Nobel Prize in Chemistry (1911): Awarded the Nobel Prize in Chemistry for the discovery of polonium and radium and the isolation of radium. She is the only person to win Nobel Prizes in two scientific fields.
- Radium Institute: She established the Radium Institute (now the Curie Institute) in Paris, a major center for research in radioactivity.
- World War I: During World War I, Marie Curie developed mobile X-ray units (“petites Curies”) to help diagnose injuries in field hospitals. She and her daughter, Irene, trained technicians to operate the units.
- Later Years: Continued her research and oversaw the Radium Institute. She became an international celebrity and a role model for women in science.
- Health Effects of Radiation: Her long-term radiation exposure (the dangers of which were not fully understood at the time) took a toll on her health.
- Death (1934): Marie Curie died on July 4, 1934, from aplastic anemia, almost certainly caused by her exposure to radiation.
- Legacy:
- Pioneers of Radioactivity: Marie and Pierre Curie were pioneers in the study of radioactivity, making fundamental discoveries that transformed physics and chemistry.
- New Elements: Their discovery of polonium and radium expanded the periodic table and opened up new avenues of research.
- Atomic Structure: Their work challenged the prevailing view of the atom as indivisible and paved the way for the development of nuclear physics.
- Medical Applications: Their discoveries had profound implications for medicine, particularly in developing radiotherapy for cancer treatment.
- Role Model for Women in Science: Marie Curie became an international icon and a role model for women in science, demonstrating that women could make groundbreaking contributions to scientific research.
- Curie Institute: The Curie Institute remains a leading research center.
- Family of Scientists: Her daughter and son-in-law would also receive the Nobel Prize.
The Curies’ story is one of extraordinary scientific achievement, personal dedication, and tragic consequences. Their collaborative work revolutionized our understanding of matter and energy, and their legacy continues to inspire scientists and researchers worldwide. Marie Curie, in particular, is one of the most important and influential scientists of all time.
Marie Curie, Radioactivity, and Legacy. Table

Caricature of Marie and Pierre Curie, captioned “Radium,” in the London magazine Vanity Fair, December 1904
(Wiki Image By Julius Mendes Price – Published in Vanity Fair, 22 December 1904.Downloaded from Science Museum, Public Domain, https://commons.wikimedia.org/w/index.php?curid=12440911)
Let’s summarize Marie and Pierre Curie’s story, their work on radioactivity, and their lasting legacy in a comprehensive table. This will encompass their scientific discoveries, personal lives (briefly), and the broader impact of their work.
| Aspect | Details |
| Early Lives (Separate) | Marie Skłodowska (1867-1934): Born in Warsaw, Poland (then part of the Russian Empire). Faced barriers to higher education as a woman. She moved to Paris in 1891 to study physics and mathematics at the Sorbonne. <br> Pierre Curie (1859-1906): Born in Paris, France. Early research on magnetism and piezoelectricity. |
| Partnership (1895-1906) | Marriage (1895): Formed a scientific and personal partnership. <br> Shared Laboratory: Worked in a poorly equipped laboratory at Paris’s School of Physics and Chemistry. |
| Discovery of Radioactivity | Becquerel’s Discovery (1896): Inspired Marie Curie to investigate uranium rays. <br> Radioactivity (1898): Marie Curie coined the term “radioactivity.” <br> Polonium and Radium (1898): Discovered two new radioactive elements, polonium and radium, through painstaking chemical separation of pitchblende. <br> Atomic Property: Marie Curie hypothesized that radioactivity was an atomic property, not a chemical one. |
| Recognition and Awards | Nobel Prize in Physics (1903): Shared with Henri Becquerel for their work on radioactivity. Marie Curie was the first woman to win a Nobel Prize. |
| Pierre’s Death (1906) | Killed in a street accident in Paris. |
| Marie Curie’s Solo Work | Professor at the Sorbonne: Took over Pierre’s professorship, becoming the first woman to hold this position. <br> Isolation of Radium (1910): Isolated pure metallic radium. <br> Nobel Prize in Chemistry (1911): Awarded for the discovery of polonium and radium and the isolation of radium. |
| World War I | Mobile X-ray Units (“petites Curies”): Developed mobile X-ray units for field hospitals. <br> Training Technicians: Trained technicians to operate the X-ray units. |
| Radium Institute | Establishment: Oversaw the establishment of the Radium Institute (now the Curie Institute) in Paris, a major center for research in radioactivity. |
| Later Life and Death | Continued Research: Continued her research and oversaw the Radium Institute. <br> Health Effects: Suffered from the long-term effects of radiation exposure. <br> Death (1934): Died of aplastic anemia, almost certainly caused by radiation exposure. |
| Legacy | Pioneers of Radioactivity: Revolutionized physics and chemistry with their discoveries. <br> New Elements: Discovered polonium and radium. <br> Atomic Structure: Challenged the idea of the atom as indivisible. <br> Medical Applications: Their work led to the development of radiotherapy for cancer treatment. <br> Role Model: Marie Curie became an international icon and a role model for women in science. <br> Curie Institute: Remains a leading research center. <br> Family Legacy: Her daughter Irène Joliot-Curie and son-in-law Frédéric Joliot-Curie also won a Nobel Prize in Chemistry (1935) for their discovery of artificial radioactivity. |
| Key Experiments | Analysis of Becquerel’s rays. Chemical separation of pitchblende. |
Export to Sheets
Key Themes:
- Scientific Collaboration: The importance of teamwork and complementary skills in scientific research.
- Perseverance: The Curies’ dedication and perseverance in the face of difficult working conditions and limited resources.
- Groundbreaking Discoveries: Their discoveries fundamentally changed our understanding of matter and energy.
- Impact on Science and Medicine: Their work profoundly impacted physics, chemistry, and medicine.
- Marie Curie’s Achievements: Her remarkable achievements as a woman in science in a male-dominated era.
This table provides a comprehensive overview of Marie and Pierre Curie’s lives, their work on radioactivity, and their lasting legacy. It highlights their contributions, collaborative partnership, and the profound impact of their discoveries on science and society. The table format allows for a clear and organized presentation of this important story.
Curie New Elements: Discovered polonium and radium. Table
Let’s present the discovery of polonium and radium by Marie and Pierre Curie in a detailed table format. This will cover the context, their methods, observations, the properties of the new elements, and the significance of their discovery.
| Aspect | Details |
| Context (Background) | – Henri Becquerel’s Discovery (1896): Uranium salts emit rays that can penetrate opaque materials. <br> – Marie Curie’s Doctoral Research: Chose to investigate these “uranic rays” (later named radioactivity). <br> – Existing Knowledge: Atoms were considered indivisible and unchanging; the nature of Becquerel’s rays was unknown. |
| Initial Observations | – Radioactivity is Atomic: Marie Curie found that the intensity of radiation from uranium compounds depended only on the amount of uranium, not its chemical form. This suggested radioactivity was an atomic property. <br> – Thorium’s Radioactivity: She discovered that thorium also emitted similar rays. <br> – Pitchblende Anomaly: She found that pitchblende (a uranium ore) was more radioactive than pure uranium, suggesting the presence of other, more radioactive elements. |
| Hypothesis | – Pitchblende contains one or more unknown elements that are more radioactive than uranium. |
| Experimental Methods | – Chemical Separation: Painstaking chemical separation techniques (fractional crystallization, precipitation) were used to isolate the radioactive components of pitchblende. This involved processing tons of ore in poor laboratory conditions. <br> – Electrometer Measurements: Used an electrometer (developed by Pierre and his brother) to quantitatively measure the intensity of radiation emitted by different fractions. This was crucial for tracking the radioactive substances. |
| Discovery of Polonium (July 1898) | – Isolation: Isolated a fraction from pitchblende that was hundreds of times more radioactive than uranium. <br> – Chemical Properties: Found that this fraction had chemical properties similar to bismuth. <br> – Name: Named the new element “polonium” after Marie Curie’s native Poland. <br> – Publication: Published their findings in Comptes Rendus de l’Académie des Sciences. |
| Discovery of Radium (December 1898) | – Further Separation: Continued to process pitchblende and isolated another fraction that was even more radioactive than polonium. <br> – Chemical Properties: Found that this fraction had chemical properties similar to barium. <br> – Spectroscopic Analysis: Obtained a new spectral line (using spectroscopy) that did not belong to any known element, confirming the presence of a new element. <br> – Name: Named the new element “radium” from the Latin word for “ray.” <br> – Publication: Published their findings in Comptes Rendus. |
| Properties of Polonium and Radium | – Extremely Radioactive: Both elements were far more radioactive than uranium. <br> – Spontaneous Emission of Radiation: They emitted radiation spontaneously, without any external energy input. <br> – Heat and Light Emission: Radium emitted heat and light. <br> – Ionizing Air: The radiation ionizes the surrounding air, making it electrically conductive. <br> – Physiological Effects: The Curies (unknowingly) experienced the harmful effects of radiation (burns, later health problems). |
| Further Work (After 1898) | – Isolation of Pure Radium: Marie Curie continued to work for years to isolate pure metallic radium (achieved in 1910). <br> – Determination of Atomic Weights: They worked to determine the atomic weights of polonium and radium. |
| Significance | – Discovery of New Elements: Expanded the periodic table. <br> – Confirmation of Radioactivity: Demonstrated that radioactivity was a phenomenon associated with multiple elements. <br> – Challenge to Atomic Theory: Challenged the prevailing view of the atom as indivisible and unchanging. <br> – Opening of New Fields: Opened up the fields of nuclear physics and radiochemistry. <br> – Medical Applications: Led to the development of radiotherapy for cancer treatment. <br> – Nobel Prizes: Recognition of the groundbreaking nature of their work. |
Export to Sheets
Key Takeaways:
- Hypothesis-Driven Research: Marie Curie’s initial hypothesis (that pitchblende contained unknown radioactive elements) guided her research.
- Painstaking Experimentation: The discovery of polonium and radium required years of arduous chemical separation and analysis, processing tons of ore.
- Quantitative Measurement: The use of the electrometer to quantify radioactivity was crucial for tracking the new elements.
- Collaboration: The discovery was a result of the close collaboration between Marie and Pierre Curie.
- Revolutionary Impact: Their discoveries revolutionized physics and chemistry, challenging existing theories and opening up new fields of research.
This table provides a detailed and structured overview of the discovery of polonium and radium by Marie and Pierre Curie, emphasizing their experimental methods, observations, and the profound significance of their work. It highlights their scientific process and the revolutionary impact of their discoveries on science and medicine.
Curie Atomic Structure: Challenged the idea of the atom as indivisible. Table
Let’s create a table that specifically focuses on how Marie and Pierre Curie’s work on radioactivity challenged the prevailing understanding of atomic structure in the late 19th and early 20th centuries, and how their discoveries contributed to the development of a new model of the atom.
| Aspect | Prevailing View of the Atom (Late 19th Century) | Curie’s Discoveries and Observations | How Curie’s Work Challenged the Prevailing View | Impact on Atomic Theory |
| Nature of the Atom | – Atoms were considered the smallest, indivisible, and unchanging units of matter. <br> – Dalton’s atomic theory (early 19th century) was widely accepted. | – Radioactivity as an atomic phenomenon: Marie Curie showed that radioactivity was a property of the atom itself, not a result of chemical reactions or external influences. <br> – Spontaneous Emission of Radiation: Radioactive elements emitted radiation spontaneously and continuously, without any external energy input. <br> – Discovery of New Elements: Polonium and radium were new elements with extremely high radioactivity. | – Implied that atoms were not indivisible: The spontaneous emission of radiation suggested that atoms were changing and emitting particles, implying that they had internal structure. <br> – Challenged the idea of atomic stability: Radioactive atoms were clearly not unchanging. | – Opened the door to the investigation of the internal structure of the atom. <br> – Led to the development of nuclear physics. <br> – Forced a fundamental rethinking of the nature of matter. |
| Energy Source | – The source of energy in chemical reactions was understood in terms of chemical bonds. <br> – No known source of energy could explain the continuous emission of heat by radium. | – Radium continuously emits heat and light without any apparent external source of energy. <br> – The amount of energy released by radioactive decay was far greater than could be explained by any known chemical process. | – Suggested a vast, previously unknown source of energy within the atom. This was a major puzzle that could not be explained by classical physics. | – Paved the way for the understanding of nuclear energy. <br> – Einstein’s E=mc² (1905) later provided the theoretical explanation: the conversion of a small amount of mass into a large amount of energy. |
| Transmutation of Elements | – Elements were considered to be immutable and unchanging. One element could not be transformed into another. (This was a core tenet of Dalton’s atomic theory). | – The Curies observed that radioactive elements emitted different types of radiation (later identified as alpha, beta, and gamma rays). <br> – They observed that the properties of radioactive materials changed over time. | – Implied that radioactive decay involved the transformation of one element into another. This was a radical idea that contradicted the long-held belief in the immutability of elements. | – Led to the understanding of radioactive decay as a process where an atom’s nucleus changes, emitting particles and energy, and transforming into a different element. <br> – Birth of nuclear chemistry. |
| Nature of Radiation | – The nature of Becquerel’s “uranic rays” was unknown. | – The Curies studied the properties of the radiation emitted by radioactive elements: <br> – Ionizing air. <br> – Affecting photographic plates. <br> – Causing fluorescence. | – Helped to characterize the different types of radiation. (Later identified as alpha, beta, and gamma rays by Rutherford and others). | – Provided the tools for further investigation of radioactivity and atomic structure. |
| Atomic Model | The Daltonian model of the atom, as a solid indivisible sphere. | The work showed this model to be false. | Atoms must have a structure. | – |
Export to Sheets
Key Takeaways:
- Revolutionary Discoveries: The Curies’ work on radioactivity was revolutionary, challenging fundamental assumptions about the nature of matter and energy.
- Experimental Evidence: Their discoveries were based on meticulous experimental work and careful observation.
- Shift in Paradigm: They initiated a paradigm shift in physics and chemistry, leading to the development of nuclear physics and a new understanding of the atom.
- Foundation for Future Research: Their work laid the foundation for much of 20th-century physics, including the discovery of the neutron, nuclear fission, and the development of nuclear weapons and nuclear power.
This table clearly shows how Marie and Pierre Curie’s research on radioactivity directly challenged the prevailing view of the atom as indivisible and unchanging, paving the way for a new understanding of atomic structure and the development of nuclear physics. It emphasizes the evidence from their experiments and how this evidence contradicted existing theories.
Curie Medical Applications: Their work led to the development of radiotherapy for cancer treatment. Table
Marie and Pierre Curie’s groundbreaking research into radioactivity had a profound impact on medicine, most notably in the development of radiotherapy for cancer treatment. Here’s a table outlining their contributions and the significance of their work:
Curie Medical Applications: Radiotherapy
| Aspect | Description | Significance |
| Discovery of Radium | * The Curies discovered radium, an intensely radioactive element, in 1898. <br> * They observed its ability to destroy diseased cells. | * Provided a powerful tool for the treatment of cancer. <br> * Opened the door to the field of radiation therapy. <br> * Radium’s properties allowed for targeted destruction of tumors. |
| Early Radiotherapy | * Pierre Curie conducted experiments, including on himself, to observe the physiological effects of radiation. <br> * They observed that tumors shrank when exposed to radium. | * Laid the foundation for the development of targeted radiation therapy. <br> * Showed that radiation could selectively destroy cancerous cells while minimizing damage to healthy tissue (to some extent, given the limited early knowledge). |
| Development of Radiotherapy Techniques | * During World War I, Marie Curie and her daughter Irene developed mobile X-ray units (“petites Curies”) to help doctors diagnose and treat wounded soldiers. <br> * These units helped locate shrapnel and fractures and reduced the need for exploratory surgeries. <br> * Radium needles were used to treat tumors. | * Greatly improved wartime medical care. <br> * Demonstrated the practical applications of radiation in medical diagnosis and treatment. <br> * Increased access to x-ray technology on the front lines. |
| Legacy | Continued research into radioactive materials, including the use of radon gas for pain relief. Curie’s work paved the way for modern radiation treatment. | Continued development of radiation therapy, including the use of radon gas and, later, cobalt-60. Modern radiation therapy saves countless lives. |
Export to Sheets
Key Points:
- Pioneering Work:
- The Curies’ work established the foundation for radiation therapy as a powerful cancer treatment.
- Direct Application:
- They actively applied their discoveries to develop practical medical technologies.
- Lasting Impact:
- Their contributions continue to save lives through modern radiation therapy.
- Wartime Applications:
- Their X-ray units played a vital role in improving medical care during World War I.
Curie Pioneers of Radioactivity: Revolutionized physics and chemistry with their discoveries. Table
The Curies’ work was truly transformative, ushering in the era of radioactivity and reshaping our understanding of the atom. Here’s a table summarizing their pioneering contributions:
**Curie Pioneers of Radioactivity
| Contribution | Description | Significance |
| Discovery of Radioactivity | * Marie Curie coined the term “radioactivity” to describe the spontaneous emission of radiation by certain elements. <br> * They showed that radioactivity is an atomic property. | * Challenged the long-held belief that atoms were indivisible. <br> * Opened a new field of physics and chemistry. <br> * Transformed the understanding of atomic structure. |
| Discovery of Polonium and Radium | * Working with Pitchblende, they identified and isolated two new radioactive elements. <br> * Polonium, named after Marie’s native Poland, and Radium, meaning “ray.” | * Significantly expanded the periodic table. <br> * Provided intensely radioactive sources for further research and medical applications. <br> * This work showed that more elements existed that were highly radioactive. |
| Investigation of Radioactive Properties | * Conducted extensive studies on the properties of radioactive substances. <br> * Measured the radiation emitted by different elements and compounds. | * Provided the data needed to understand the nature of radioactive decay. <br> * Laid the foundation for future studies of nuclear physics. <br> * Their work created a base that other scientists could then build upon. |
| Development of Experimental Techniques | * Pioneered methods for isolating and measuring radioactive materials. <br> * Designed sensitive instruments to detect and quantify radiation. | * Made it possible to conduct accurate studies of radioactivity. <br> * These techniques were crucial for advancing the field. <br> * By creating ways to measure radioactivity, they gave themselves the tools to create more discoveries. |
Export to Sheets
Key Points:
- Paradigm Shift:
- The Curies’ work caused a fundamental shift in scientific thinking about the nature of matter.
- Expansion of Knowledge:
- Their discoveries significantly expanded the known elements and properties of matter.
- Foundation for Nuclear Physics:
- Their research laid the groundwork for the development of nuclear physics and its various applications.
- Two Nobel Prizes:
- Marie Curie made history by being the first woman to win a Nobel Prize, and also being the first person to win two Nobel Prizes.
Marie Curie Role Model: She became an international icon and a role model for women in science. Table
Marie Curie’s life and work have had a profound impact that extends far beyond her scientific discoveries. She became a symbol of perseverance, dedication, and the power of women in science. Here’s a table highlighting her role as a role model:
Marie Curie: Role Model for Women in Science
| Aspect | Description | Significance |
| Breaking Barriers | * Overcame significant social and gender barriers to pursue her scientific education and career. <br> * Achieved recognition and success in a field dominated by men. | * Demonstrated that women could excel in science at the highest levels. <br> * Paved the way for future generations of women in STEM. <br> * Her success challenged societal norms and expectations. |
| Dedication and Perseverance | * Showed unwavering dedication to her research, even in the face of hardship and adversity. <br> * Continued her work despite limited resources and health challenges. | * Inspired others to pursue their passions with determination and resilience. <br> * Reinforced the importance of perseverance in scientific discovery. <br> * A powerful example of pursuing one’s dreams regardless of difficulty. |
| Scientific Excellence | * Achieved unparalleled scientific success, winning two Nobel Prizes in different fields. <br> * Her rigorous research and groundbreaking discoveries set a high standard for scientific excellence. | * Provided a powerful example of scientific excellence and innovation. <br> * Elevated the perception of women’s capabilities in scientific research. <br> * Demonstrated that women are capable of the same level of intellectual achievement as men. |
| Humanitarian Spirit | * Applied her scientific knowledge to improve medical care, particularly during World War I. <br> * Developed mobile X-ray units to help wounded soldiers. | * Showed the importance of using science for the benefit of humanity. <br> * Emphasized the ethical responsibility of scientists. <br> * Created an image of the scientist as someone who uses their knowledge to directly improve the lives of others. |
| Inspiring Future Generations | The stories of her achievements continue to motivate girls and women around the globe to pursue careers in science. | Her legacy extends beyond her discoveries by inspiring countless people. |
Export to Sheets
Key Points:
- Symbol of Empowerment:
- Marie Curie became a symbol of empowerment for women worldwide.
- Overcoming Obstacles:
- Her life story demonstrated that women could overcome significant obstacles to achieve scientific success.
- Lasting Legacy:
- She left a lasting legacy that continues to inspire and motivate women in science.
Similarities Antoine Lavoisier, John Dalton, Jöns Jacob Berzelius, Dmitri Mendeleev, and Marie Curie
While Antoine Lavoisier, John Dalton, Jöns Jacob Berzelius, Dmitri Mendeleev, and Marie Curie made distinct contributions to different areas of chemistry and lived in other eras, there are several significant similarities in their approaches, their impact, and their legacies:
- Emphasis on Quantitative Experimentation and Observation:
- All five were meticulous experimentalists (or, in Mendeleev’s case, a brilliant synthesizer of experimental data). They emphasized precise measurement and careful observation as the foundation for chemical knowledge. This contrasted with earlier, more speculative or qualitative approaches.
- Lavoisier: Revolutionized chemistry by introducing quantitative methods and the law of conservation of mass.
- Dalton: Based his atomic theory on quantitative observations of combining ratios of elements.
- Berzelius: Devoted years to determining accurate atomic weights through painstaking experiments.
- Mendeleev: Used quantitative data on atomic weights and properties to construct his periodic table.
- Curie: Pioneered precise measurements of radioactivity and developed techniques for isolating radioactive elements.
- Development of Fundamental Chemical Concepts:
- All five contributed to developing fundamental concepts still central to chemistry today. They didn’t just make isolated discoveries; they built frameworks for understanding.
- Lavoisier: Oxygen theory of combustion, law of conservation of mass, elements vs. compounds.
- Dalton: Atomic theory (matter is composed of atoms).
- Berzelius: Chemical notation, accurate atomic weights, concepts of isomerism and catalysis.
- Mendeleev: Periodic law and the periodic table of elements.
- Curie: Radioactivity (challenging the indivisibility of the atom).
- Systematization and Organization of Chemical Knowledge:
- All five contributed to the systematization and organization of chemical knowledge, making it more coherent and accessible.
- Lavoisier: New chemical nomenclature.
- Dalton: Atomic theory provided a unifying framework.
- Berzelius: Chemical notation, systematization of analytical techniques, annual reports on chemical progress.
- Mendeleev: The periodic table provided a comprehensive organizing system for the elements.
- Curie: Defined and named Radioactivity.
- Challenging Existing Theories:
- All five challenged existing theories or paradigms in chemistry and physics. They were not afraid to question established ideas and to propose new ones based on evidence.
- Lavoisier: Overthrew the phlogiston theory.
- Dalton: Challenged the idea of matter as a continuum.
- Mendeleev: Challenged the notion that atomic weight was the sole basis for classifying elements.
- Curie: Challenged the idea of the atom as indivisible and unchanging.
- Lasting Impact on Chemistry and Science:
- Their work had a profound and lasting impact on the development of chemistry and other scientific fields. Their discoveries and theories are still taught today, and their methods influence scientific research.
- Lavoisier: Foundation of modern quantitative chemistry.
- Dalton: Atomic theory is the basis for understanding chemical reactions.
- Berzelius: Chemical notation and fundamental concepts still in use.
- Mendeleev: Periodic table as a cornerstone of chemistry.
- Curie: Opened up the field of nuclear physics and radiochemistry.
In summary: These five chemists, despite their differences in background, specialization, and historical context, share a common thread: they were all transformative figures in the history of chemistry. They emphasized quantitative experimentation, developed fundamental concepts, systematized chemical knowledge, challenged existing theories, and left a lasting legacy that continues to shape the field today. They represent the power of scientific inquiry to unlock the secrets of the natural world. They built upon each other’s work, creating a continuous (though not always linear) progression of chemical knowledge.
Differences of Antoine Lavoisier, John Dalton, Jöns Jacob Berzelius, Dmitri Mendeleev, and Marie Curie
While the previous response highlighted the similarities, it’s equally important to understand the differences between Antoine Lavoisier, John Dalton, Jöns Jacob Berzelius, Dmitri Mendeleev, and Marie Curie. These differences encompass their historical context, specific areas of focus within chemistry, methodologies, and the nature of their discoveries.
- Historical Context and Era:
- Lavoisier (1743-1794): Late 18th century – Enlightenment and the French Revolution. The transition from alchemy to more quantitative chemistry.
- Dalton (1766-1844): Late 18th and early 19th century – Industrial Revolution in England. Early development of atomic theory.
- Berzelius (1779-1848): First half of the 19th century – Consolidation of chemical knowledge; development of analytical techniques.
- Mendeleev (1834-1907): Mid-to-late 19th century – Rise of systematic inorganic chemistry; an increasing number of known elements.
- Curie (1867-1934): Late 19th and early 20th century – Discovery of radioactivity; dawn of nuclear physics.
This difference in time period is crucial. Each chemist built upon the work of their predecessors, and the tools and knowledge available to them changed significantly.
- Primary Area of Focus within Chemistry:
- Lavoisier: Combustion, respiration, chemical nomenclature, the composition of air and water, the law of conservation of mass. The foundation of quantitative chemistry.
- Dalton: Atomic theory, gas laws, relative atomic weights. Theoretical framework for understanding matter.
- Berzelius: Chemical notation, precise determination of atomic weights, discovery of elements, electrochemical theory, isomerism, catalysis. Systematization and analytical chemistry.
- Mendeleev: Organization of the elements, periodic law, prediction of undiscovered elements. Inorganic chemistry and the relationships between elements.
- Curie: Radioactivity, discovery, and isolation of radioactive elements (polonium and radium). Nuclear physics and radiochemistry.
- Methodology and Approach:
- Lavoisier: Meticulous quantitative experimentation, focusing on mass measurements in closed systems. Strong emphasis on experimentally disproving existing theories (phlogiston).
- Dalton: More theoretical, developing his atomic theory based on existing experimental data (laws of definite and multiple proportions) and his gas studies. Less emphasis on discovering new phenomena and more on explaining existing ones.
- Berzelius: Highly skilled experimentalist and analyst. Focused on precise measurements, developing new analytical techniques, and systematizing chemical knowledge.
- Mendeleev: Primarily a theorist and synthesizer. He used existing data on element properties and atomic weights to create his periodic table, making predictions based on patterns.
- Curie: Primarily experimental, focused on isolating and characterizing new radioactive elements. Pioneering and physically demanding experimental work.
- Nature of Discoveries:
- Lavoisier: Revolutionary discoveries about the nature of combustion and the role of oxygen; established fundamental laws.
- Dalton: Proposed a fundamental theory (atomic theory) that explained existing observations.
- Berzelius: Made numerous incremental contributions that systematized and refined chemical knowledge; discovered new elements and concepts.
- Mendeleev: Created a comprehensive organizing system (periodic table) that had predictive power.
- Curie: Discovered a completely new phenomenon (radioactivity) and new elements, opening up a new field of science.
- Social and Political Context:
- Lavoisier: French aristocracy; involved in pre-revolutionary and early revolutionary government; executed during the Reign of Terror.
- Dalton: English Quaker; modest background; primarily a teacher and researcher.
- Berzelius: Swedish; became a dominant figure in European chemistry; established a strong scientific tradition in Sweden.
- Mendeleev: Russian; worked within the context of a rapidly changing Russian society; faced some resistance to his ideas initially.
- Curie: Polish-French; faced significant gender barriers as a woman in science; became an international celebrity.
- Relationship to Physics:
- Lavoisier, Dalton, Berzelius: Primarily chemists, though their work had implications for physics.
- Mendeleev: Primarily a chemist, though the periodic table ultimately found its explanation in atomic physics.
- Curie: Her work on radioactivity bridged chemistry and physics, opening up the field of nuclear physics.
In summary, while all five figures were crucial to the development of chemistry, they made distinct contributions in different areas, using different methodologies and working within different historical and social contexts. Lavoisier laid the quantitative foundations, Dalton provided the atomic framework, Berzelius systematized chemical knowledge, Mendeleev organized the elements, and Curie opened up the field of radioactivity. Understanding these differences is as important as recognizing their shared commitment to scientific inquiry. They represent a progression of ideas and discoveries, each building upon (and sometimes challenging) the work of their predecessors.
Compared to Antoine Lavoisier, John Dalton, Jöns Jacob Berzelius, Dmitri Mendeleev, and Marie Curie
You’re asking for a comparison of those five chemists to each other. The previous response provided a detailed comparison between them. This response will summarize each person’s relative strengths, weaknesses, and styles compared to the group’s others. This is inherently subjective, but I’ll base it on their historical impact and the nature of their contributions.
- Antoine Lavoisier (1743-1794):
- Strengths:
- Revolutionary Experimentalist: His meticulous quantitative experiments and focus on closed systems were groundbreaking.
- Clear Thinker: He had a remarkable ability to design experiments that directly tested existing theories (phlogiston) and to interpret his results logically.
- Communicator: His Traité Élémentaire de Chimie was clear and systematic, effectively disseminating his new chemistry.
- Systematizer: Developed a new chemical nomenclature.
- Weaknesses:
- Less of a Theorist (compared to Dalton): While he developed the oxygen theory, he didn’t propose a comprehensive atomic theory like Dalton.
- Limited Scope (compared to Mendeleev): His work focused on specific reactions and substances, not the overall organization of all elements.
- Incorrect on Acids: His belief that all acids contained oxygen was a significant error (though a productive one in driving further research).
- Style: Rigorous, quantitative, experimental, focused on disproving existing theories.
- John Dalton (1766-1844):
- Strengths:
- Theoretical Vision: Developed the foundational atomic theory that unified and explained many chemical observations.
- Simplicity and Explanatory Power: His theory was remarkably simple yet explained many phenomena.
- Quantitative Reasoning: He connected his theory to quantitative relationships (combining weights).
- Weaknesses:
- Less Skilled Experimentalist (compared to Lavoisier or Berzelius): His experimental work was less sophisticated than that of Lavoisier or Berzelius. He relied heavily on existing data.
- Inaccurate Atomic Weights: His initial atomic weights were often imprecise due to limitations in experimental techniques and some incorrect assumptions.
- Less Comprehensive System (compared to Mendeleev): His theory explained how atoms combine but didn’t provide a system for organizing all elements.
- Style: Theoretical, based on existing experimental data, focused on fundamental principles.
- Jöns Jacob Berzelius (1779-1848):
- Strengths:
- Master Experimentalist and Analyst: He was renowned for his meticulous experimental skills and his development of new analytical techniques.
- Systematizer: He brought order and precision to chemical notation, terminology, and data (atomic weights).
- Discoverer of Elements: Discovered several new elements.
- Influential Teacher and Writer: His textbooks and annual reports were highly influential.
- Weaknesses:
- Less of a Revolutionary Theorist (compared to Dalton or Lavoisier): His theoretical contributions (electrochemical dualism) were less transformative than Dalton’s atomic theory or Lavoisier’s oxygen theory.
- Dogmatic: He could be dogmatic and resistant to new ideas that challenged his views.
- Style: Meticulous experimentalist, systematizer, and influential figure in the chemical community.
- Dmitri Mendeleev (1834-1907):
- Strengths:
- Visionary Organizer: He saw the pattern in the properties of the elements that others had missed, creating the periodic table.
- Predictive Power: His table didn’t just organize existing knowledge; it predicted the existence and properties of undiscovered elements.
- Boldness: He was willing to leave gaps in his table and to deviate from strict atomic weight orders when necessary.
- Weaknesses:
- Less of an Experimentalist (compared to Lavoisier or Berzelius): His greatest contribution was organizing existing data, not conducting groundbreaking experiments himself (though he was a capable experimentalist).
- Initial Resistance: His table faced some initial resistance from the scientific community.
- Underlying Explanation Lacking (initially): He couldn’t explain why the periodic law worked (that came later with atomic structure).
- Style: Synthesizer, organizer, predictor; saw patterns and relationships that others missed.
- Marie Curie (1867-1934):
- Strengths:
- Pioneering Experimentalist: Pioneered the study of radioactivity, a completely new phenomenon.
- Perseverance: Her work involved incredibly painstaking and physically demanding chemical separations.
- Discoverer of Elements: Discovered two new elements (polonium and radium).
- Challenged Existing Paradigms: Her work challenged the idea of the atom as indivisible and unchanging.
- Weaknesses:
- Less of a Theorist (compared to Dalton): Her primary contributions were experimental discoveries, though her work had profound theoretical implications.
- Health Risks: She was unaware of the long-term health risks of radiation exposure, which ultimately led to her death.
- Style: Determined, meticulous experimentalist; opened up a new field of science (nuclear physics).
In relative terms:
- Lavoisier: The experimental revolutionary who overthrew an old paradigm.
- Dalton: The theoretical architect who provided the atomic foundation.
- Berzelius: The meticulous experimentalist and systematizer who refined and organized chemical knowledge.
- Mendeleev: The visionary who saw the overarching pattern in the elements.
- Curie: The pioneer who opened up a new field of science (radioactivity) through groundbreaking experimental work.
They all built upon each other’s work (though not always directly or consciously). Lavoisier’s quantitative approach was essential for Dalton’s atomic theory. Dalton’s theory provided the framework that Berzelius refined with his precise measurements. Mendeleev’s table was based on the accumulated knowledge of element properties and atomic weights (to which Berzelius was a major contributor). Curie’s work challenged the Daltonian view of the atom as indivisible, ushering in the era of nuclear physics. They represent a progression of scientific understanding, each making unique and essential contributions.



