Gilbert N. Lewis, Linus Pauling, Dorothy Crowfoot Hodgkin, Robert Burns Woodward, and Ahmed Zewail: Modern Chemists
Let’s delve into the lives, key contributions, and significance of Gilbert N. Lewis, Linus Pauling, Dorothy Crowfoot Hodgkin, Robert Burns Woodward, and Ahmed Zewail – five outstanding chemists who made groundbreaking contributions to different areas of chemistry.
- Gilbert N. Lewis (1875-1946):
- Nationality: American
- Key Contributions:
- Covalent Bond (1916): Proposed the covalent bond concept, where atoms share electrons to achieve stability rather than transferring them (as in ionic bonds). This was a fundamental advance in understanding chemical bonding. He introduced the idea of electron pairs.
- Lewis Structures (Electron Dot Diagrams): Developed Lewis structures, a simple and widely used way to represent the valence electrons and bonding in molecules. These diagrams are a staple of introductory chemistry.
- Lewis Acids and Bases (1923): Defined acids and bases regarding electron-pair donation and acceptance, broadening the definition beyond proton donors and acceptors (Brønsted-Lowry definition). A Lewis acid accepts an electron pair, and a Lewis base donates an electron pair.
- Thermodynamics: Made significant contributions to chemical thermodynamics, applying thermodynamic principles to chemical systems.
- Photon: Coined the name.
- Heavy Water: Isolated pure heavy water.
- Significance: Lewis’s work on chemical bonding is fundamental to modern chemistry. His concept of the covalent bond and Lewis structures is an essential tool for understanding and predicting the behavior of molecules. His acid-base theory is also widely used. He was nominated for the Nobel Prize 41 times but, surprisingly, never received it, a fact often cited as a significant oversight.
- Linus Pauling (1901-1994):
- Nationality: American
- Key Contributions:
- Quantum Chemistry: One of the founders of quantum chemistry, who applied the principles of quantum mechanics to explain the nature of the chemical bond. He developed the valence bond theory, a quantum mechanical description of bonding.
- Electronegativity: Introduced the concept of electronegativity, a measure of an atom’s ability to attract electrons in a chemical bond. He developed the Pauling electronegativity scale.
- Structure of Proteins: Determined the alpha-helix and beta-sheet structures, two fundamental structural elements of proteins. This was a breakthrough in understanding the complex structures of biological molecules.
- Molecular Biology: He contributed significantly to understanding the structure and function of biological molecules, including DNA (though he did not discover the double helix structure, his work was influential).
- Hybridization: Atomic orbital hybridization.
- Resonance:
- Activism: A prominent advocate for nuclear disarmament and peace, receiving the Nobel Peace Prize in 1962.
- Significance: Pauling was one of the most influential chemists of the 20th century. His work on chemical bonding revolutionized our understanding of how atoms combine to form molecules. His structural studies of proteins were crucial for the development of molecular biology. He is the only person to have won two unshared Nobel Prizes (Chemistry in 1954 and Peace in 1962).
- Dorothy Crowfoot Hodgkin (1910-1994):
- Nationality: British
- Key Contributions:
- X-ray Crystallography: Pioneered X-ray crystallography to determine the three-dimensional structures of complex molecules. This technique involves analyzing the diffraction patterns produced when X-rays are passed through a crystal.
- Structure of Penicillin (1945): The structure of penicillin was determined, a crucial breakthrough that enabled its mass production during World War II.
- Structure of Vitamin B12 (1956): She determined the structure of vitamin B12, a very complex molecule, for which she received the Nobel Prize in Chemistry in 1964.
- Structure of Insulin (1969): After decades of work, she determined the structure of insulin, a hormone essential for regulating blood sugar.
- Significance: Hodgkin was a master of X-ray crystallography, and her work had a profound impact on chemistry, biology, and medicine. Determining the structures of these complex molecules was essential for understanding their function and developing new drugs. She was a role model for women in science.
- Robert Burns Woodward (1917-1979):
- Nationality: American
- Key Contributions:
- Organic Synthesis: Considered the preeminent organic chemist of the 20th century. He achieved the total synthesis of many complex natural products, including:
- Quinine (an anti-malarial drug)
- Cholesterol and Cortisone (Steroids)
- Chlorophyll (the pigment responsible for photosynthesis)
- Vitamin B12 (a very complex synthesis, a collaborative effort with Albert Eschenmoser)
- Woodward-Hoffmann Rules (1965): Developed (with Roald Hoffmann) the Woodward-Hoffmann rules, which predict the stereochemistry (three-dimensional arrangement of atoms) of pericyclic reactions. These rules are based on the symmetry properties of molecular orbitals.
- Spectroscopy: Made significant contributions to using physical methods to determine chemical structure.
- Organic Synthesis: Considered the preeminent organic chemist of the 20th century. He achieved the total synthesis of many complex natural products, including:
- Significance: Woodward’s syntheses were considered works of art, demonstrating the power of organic chemistry to create complex molecules. He pushed the boundaries of what was thought possible in organic synthesis. His work significantly impacted the development of new drugs and materials. He received the Nobel Prize in Chemistry in 1965.
- Ahmed Zewail (1946-2016):
- Nationality: Egyptian-American
- Key Contributions:
- Femtochemistry: Developed femtochemistry, a field of physical chemistry that uses ultrashort laser pulses (femtoseconds – one quadrillionth of a second) to study real-time chemical reactions. This allows scientists to observe the breaking and forming of chemical bonds, something that was previously impossible.
- Revolutionary Technique: He developed techniques to use ultrashort laser pulses to observe chemical reactions in real time, essentially making “movies” of atoms and molecules as they react. This was a revolutionary breakthrough. Before Zewail, it was thought impossible to observe the actual process of bond breaking and formation.
- “Father of Femtochemistry”: He is considered the “father of femtochemistry” for his pioneering work in this field.
- Significance: Zewail’s work opened up a new way to study chemical reactions, allowing scientists to observe the fundamental processes of chemical change on an incredibly fast timescale. This has significantly impacted our understanding of chemical dynamics and has applications in many areas of chemistry and biology. He received the Nobel Prize in Chemistry in 1999.
In summary, these five chemists represent a diverse range of specializations within chemistry (physical, organic, inorganic, biochemistry, and femtochemistry) and have made groundbreaking contributions that have advanced the field significantly. They all pushed the boundaries of what was known and developed new tools and techniques that continue to be used by chemists today. Their work highlights the power of both theoretical insights and experimental skills in driving scientific progress.
Gilbert N. Lewis; Covalent Bond

Gilbert Newton Lewis
(Wiki Image By https://www.alphachisigma.org/page.aspx?pid=486, Fair use, https://en.wikipedia.org/w/index.php?curid=36700424)
Gilbert N. Lewis Quotes. Table
Let’s present key quotes from Gilbert N. Lewis in a table format, providing the source (where known), context, and significance for each.
| Quote Number | Quote | Source | Significance/Explanation |
| 1 | “Two atoms may conform to the rule of eight, or the octet rule, not only by the transfer of electrons from one atom to another, but also by sharing one or more pairs of electrons…” | Valence and the Structure of Atoms and Molecules (1923) | Defines the Covalent Bond: Explicitly states the concept of electron sharing as a way for atoms to achieve stable electron configurations (octet rule), contrasting it with ionic bonding (electron transfer). This is the foundation of his bonding theory. |
| 2 | “We are tempted to assume that the pair of electrons constitutes the chemical bond.” | “The Atom and the Molecule” (1916) | Genesis of the Shared Pair Idea: This quote from his earlier seminal paper shows the initial conceptualization of the shared electron pair as the fundamental basis of the covalent bond. |
| 3 | “[Lewis structures allow us] to visualize the arrangement of electrons in molecules and thus predict their properties.” | Representative (summarizing the purpose of Lewis structures) | Explains Lewis Structures: Highlights the purpose of his electron-dot diagrams – to provide a simple, visual model for understanding electron distribution and bonding, which helps predict molecular behavior. |
| 4 | “An acid substance is one which can employ an electron lone pair from another molecule in completing the stable group of one of its own atoms.” | Representative (paraphrasing his definition of a Lewis acid) | Defines Lewis Acids: Introduces his broader definition of acids as electron-pair acceptors, expanding acid-base chemistry beyond proton transfer. |
| 5 | “Thermodynamics predicts the possibilities, not the probabilities.” | Attributed / Representative | View on Thermodynamics: This view reflects his understanding that thermodynamics determines whether a reaction can occur spontaneously (possibility/feasibility) but not how fast it will occur (probability/rate). |
| 6 | “Science has positively demonstrated that all the energy available for human work and human life has its ultimate origin in the sun.” | Thermodynamics and the Free Energy of Chemical Substances (1923) | Broader Scientific View: From his influential thermodynamics textbook, reflecting on the ultimate energy source. |
Export to Sheets
Key Takeaways:
- Focus on Electrons: Lewis’s key conceptual breakthroughs centered on the role of valence electrons in forming chemical bonds.
- Conceptual Models: He emphasized simple, visual models (like Lewis structures) to represent complex chemical ideas.
- Broad Definitions: He sought broader, more general definitions for acids and bases.
- Foundational Impact: His ideas on bonding and structure remain fundamental to modern chemistry education and practice.
This table provides an overview of essential statements and concepts attributed to G.N. Lewis, highlighting his revolutionary contributions to chemical bonding theory and acid-base chemistry and his views on thermodynamics. The format allows for a clear understanding of the context and significance of each quote.
Gilbert N. Lewis YouTube Video
- Gilbert Newton Lewis by Carly Newman: 8,736 views (http://www.youtube.com/watch?v=m3bYNglKAwY)
- Gilbert N. Lewis: Discovering the Building Blocks of Chemistry by Laboratory Legends: 339 views (http://www.youtube.com/watch?v=UQg3unEgK1I)
- Gilbert N Lewis (EN) by about-the-world: 64 views (http://www.youtube.com/watch?v=9WuwWxH6pTk)
- Gilbert N. Lewis – A Glimpse of Motivational Wisdom #motivational #shorts #motivation #quotes by EDUwise: Engineering Knowledge & Learning: 67 views (http://www.youtube.com/watch?v=uxGsr_6RllI)
- Who was Gilbert Newton Lewis? by Real Life Top 10s: 152 views (http://www.youtube.com/watch?v=RjetyhcaTN8)
Gilbert N. Lewis History

Heavy Water
(Wiki Image By Streng – Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=35278884)
Gilbert Newton Lewis (1875-1946) was a highly influential American physical chemist. He is best known for his work on chemical bonding (the covalent bond and Lewis structures), his theory of acids and bases, and his contributions to thermodynamics. Although nominated many times, he controversially never received the Nobel Prize despite his profound impact on the field.
- Early Life and Education (1875-1904):
- Born in Weymouth, Massachusetts (1875): Grew up in Nebraska and showed early intellectual promise.
- University of Nebraska (briefly): I attended the University of Nebraska for two years.
- Harvard University (B.S., 1896; Ph.D., 1899): He transferred to Harvard, where he earned his bachelor’s and doctoral degrees in chemistry. Theodore William Richards (a Nobel laureate) supervised his doctoral research on electrochemical potentials.
- Postdoctoral Studies (Germany): He spent time studying in Germany with Walther Nernst (in Göttingen) and Wilhelm Ostwald (in Leipzig), two leading physical chemists of the time. This exposed him to the latest developments in thermodynamics and electrochemistry.
- Instructor at Harvard (1901-1903, 1904-1905): After returning from Europe, he taught at Harvard for a few years.
- Manila, Philippines (1904): Superintendent of Weights and Measures.
- MIT and Early Work on Thermodynamics (1907-1912):
- Massachusetts Institute of Technology (MIT): He joined the faculty at MIT, where he began his work on chemical thermodynamics, applying rigorous thermodynamic principles to chemical systems.
- Relativity Paper: In 1908, he published a paper on relativity.
- Focus on Thermodynamics:
- Developed methods for determining free energies of chemical substances.
- Published (with Merle Randall) Thermodynamics and the Free Energy of Chemical Substances (1923), a landmark text that greatly influenced the field.
- University of California, Berkeley (1912-1946):
- Move to Berkeley (1912): He became Dean of the College of Chemistry at the University of California, Berkeley. He transformed the chemistry department into a world-leading center for research and education.
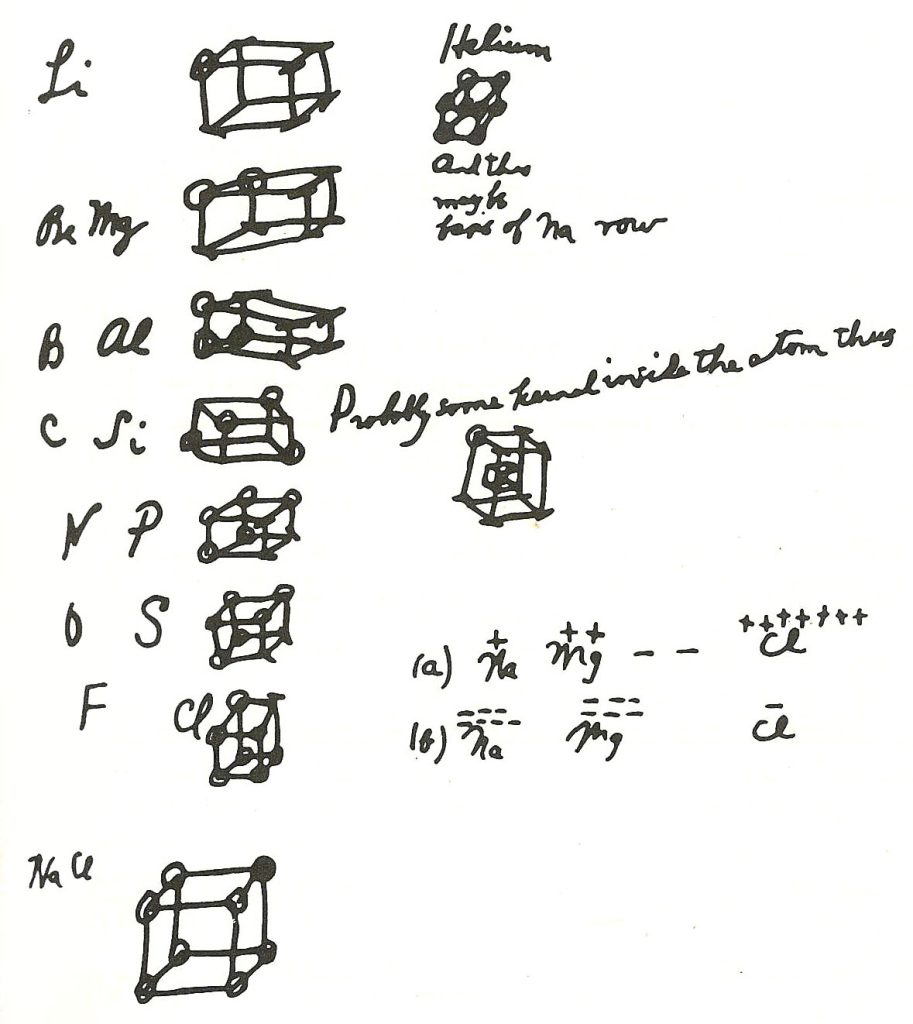
- The Covalent Bond and Lewis Structures (1916):
- “The Atom and the Molecule” (1916): He published his seminal paper proposing the concept of the shared electron pair bond (covalent bond). This revolutionary idea explained the bonding in many molecules, particularly organic molecules, where the existing ionic bonding theory didn’t work.
- Electron Pairs: He proposed that atoms share electrons to achieve a stable outer shell (usually eight electrons, the “octet rule”).
- Lewis Structures: Introduced the “cubical atom” model (a precursor to his more famous electron dot diagrams) to visualize electron sharing. While the cubical atom was incorrect, the underlying idea of shared electron pairs was groundbreaking.
- Lewis Dot Diagrams: Developed the simple and intuitive Lewis structures (electron dot diagrams) to represent the valence electrons and molecular bonding. These diagrams are still a cornerstone of chemical education.
- Lewis Acid-Base Theory (1923):
- Expanded Definition of Acids and Bases: Proposed a broader definition of acids and bases:
- Lewis Acid: An electron-pair acceptor.
- Lewis Base: An electron-pair donor.
- Significance: This definition expanded the concept of acid-base reactions beyond proton transfer (Brønsted-Lowry theory) and included a much wider range of chemical reactions.
- Expanded Definition of Acids and Bases: Proposed a broader definition of acids and bases:
- Photon (1926):
- Coined the word.
- Heavy Water (1933): * First to produce pure Heavy Water.
- Other Contributions:
- Thermodynamics: He continued his work on chemical thermodynamics, publishing influential papers and books.
- Valence and Bonding: He further refined his valence and chemical bonding theories.
- Mentorship: Mentored many students who became prominent chemists, including several Nobel laureates (Harold Urey, Glenn T. Seaborg, and others).
- Later Life and Death:
- Manhattan Project (Declined Involvement): The U.S. declined to participate in the Manhattan Project (the development of the atomic bomb) during World War II, possibly due to pacifist leanings or disagreements with the project’s leadership.
- Death (1946): He died in his laboratory at Berkeley on March 23, 1946, at 70. The circumstances of his death are somewhat mysterious; it may have been a heart attack, but some speculate it was suicide (he was reportedly depressed about a professional dispute).
- Nobel Prize Controversy Nominated many times, he never won.
- Legacy:
- He revolutionized Chemical Bonding. His concept of the covalent bond and Lewis structures is fundamental to a modern understanding of molecular structure and reactivity.
- Expanded Acid-Base Theory: His Lewis acid-base theory is widely used in organic and inorganic chemistry.
- Influential Teacher and Mentor: He trained and inspired generations of chemists at Berkeley.
- “Unrecognized” Genius: Many consider his never receiving the Nobel Prize in Chemistry a significant oversight, given the profound impact of his work. He was nominated 41 times.
- Foundation of Understanding: Made complex topics easier to understand.
Gilbert N. Lewis was one of the most important and influential American chemists of the 20th century. His contributions to chemical bonding, acid-base theory, and thermodynamics are foundational to modern chemistry. His work transformed how chemists think about molecules and chemical reactions, and his legacy continues to shape the field today. His story also reminds us that even the most outstanding scientists can be overlooked for major awards, highlighting the process’s complexities and sometimes subjective nature.
Gilbert N. Lewis, Covalent Bond, and Legacy. Table
Lewis’s cubical atoms (as drawn in 1902)
(Wiki Image PD-US, https://en.wikipedia.org/w/index.php?curid=7071913)
Let’s present the key aspects of Gilbert N. Lewis’s (1875-1946) work in a structured table format, focusing on his contributions to understanding the covalent bond and his broader legacy.
| Aspect | Details | Significance |
| Context (Pre-Lewis) | – Ionic Bonding Understood: The concept of ionic bonding (transfer of electrons between atoms) was relatively well understood, explaining the formation of salts. <br> – Covalent Bonding a Mystery: The nature of bonding in non-ionic compounds (e.g., organic molecules, diatomic gases like H₂) was unclear. Why do atoms share electrons? | – Existing bonding theories could not adequately explain the formation of many common molecules. |
| Key Contributions: Covalent Bond | – “The Atom and the Molecule” (1916): Published his seminal paper proposing the concept of the shared electron pair bond (covalent bond). <br> – Electron Sharing: Atoms can achieve stability (an entire outer electron shell, like noble gases) by sharing electrons rather than ultimately transferring them. <br> – Octet Rule: Emphasized the tendency of atoms to achieve a stable outer shell of eight electrons (or two for hydrogen). <br> – Lewis Structures (Electron Dot Diagrams): Developed a simple and visual way to represent the valence electrons and bonding in molecules, using dots to represent electrons. | – Revolutionized understanding of chemical bonding. It provided a fundamental explanation for forming molecules in many substances, particularly organic molecules. <br> – Unified chemical bonding: Showed a spectrum of bonding. <br> – Lewis structures remain a cornerstone of chemical education. |
| Key Contributions: Acids and Bases | – Lewis Acid-Base Theory (1923): Proposed a broader definition of acids and bases: <br> – Lewis Acid: An electron-pair acceptor. <br> – Lewis Base: An electron-pair donor. | – Expanded the concept of acid-base reactions beyond the traditional proton-transfer definition (Brønsted-Lowry theory). <br> – Applicable to a broader range of chemical reactions, including those that don’t involve hydrogen ions. |
| Other Contributions | – Thermodynamics: Made significant contributions to chemical thermodynamics, applying thermodynamic principles to chemical systems. <br> – “Photon” Term: Coined the term “photon” for the quantum of light (though his concept of the photon differed somewhat from the modern understanding). <br> – Heavy Water: The first isolate is a pure sample of Heavy Water. | |
| Teaching and Mentorship | – Professor at UC Berkeley: He spent most of his career as a professor at the University of California, Berkeley, building a renowned chemistry department. <br> – Mentored many students: Influenced generations of chemists, including several Nobel laureates. | – Created a major center for chemical research. <br> – Had a lasting impact on chemical education. |
| Legacy | – Foundation of Modern Bonding Theory: His work on the covalent bond is fundamental to the modern understanding of molecular structure and reactivity. <br> – Lewis Structures: His dot diagrams remain essential for chemists. <br> – Lewis Acid-Base Theory: His acid-base theory is widely used, particularly in organic and inorganic chemistry. <br> – Influence on Physical Chemistry: His work on thermodynamics and other areas had a significant impact. <br> – The “Unrecognized Nobel Laureate”: He was nominated for the Nobel Prize in Chemistry 41 times but never received it, a widely acknowledged oversight. | – His contributions are essential to understanding chemistry at a fundamental level. <br> – His work continues to be taught and used by chemists worldwide. <br> – He is considered one of the most essential American chemists. |
Export to Sheets
Key Points about Lewis’s Approach:
- Intuitive and Visual: His Lewis structures provided a simple, visual way to understand chemical bonding, making it accessible to a broader audience.
- Empirical Basis: His theories were based on experimental observations and chemical properties.
- Generalizations: He sought to develop general principles explaining various chemical phenomena.
- Bridged gaps: He showed the connection between different areas of chemistry.
This table provides a structured overview of Gilbert N. Lewis’s key contributions, emphasizing his work on the covalent bond, his broader impact on chemistry, and his lasting legacy. It highlights the revolutionary nature of his ideas and their continued relevance to modern chemistry. The format allows for a clear understanding of the context, his key achievements, and their significance.
Lewis Foundation of Modern Bonding Theory: His work on the covalent bond is fundamental to the modern understanding of molecular structure and reactivity. Table

σ bond between two atoms: localization of electron density
(Wiki Image By SVG: ZooFari; Raster: Anselm H. C. Horn – SVG work by uploader; raster by Anselm H. C. Horn, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=7050089)
You’re right to emphasize the pivotal role of Gilbert N. Lewis in shaping our understanding of chemical bonding. Here’s a table summarizing his foundational contributions:
Gilbert N. Lewis: Foundation of Modern Bonding Theory
| Contribution | Description | Significance |
| Covalent Bond | * It is proposed that the sharing of electron pairs between atoms forms chemical bonds. <br> * Developed the concept of electron sharing as a mechanism for forming stable molecules. | * Shifted the paradigm from ionic bonding to a more comprehensive understanding of molecular forces. <br> * Laid the foundation for understanding the structures and properties of vast number of compounds, especially in organic chemistry. <br> * Provided a model explaining how molecules were held together, allowing scientists to study them more deeply. |
| Lewis Dot Structures | * Introduced a simple visual representation of valence electrons using dots around atomic symbols. <br> * Illustrated how atoms share electrons to form covalent bonds. | * Provided a powerful tool for visualizing molecular structure and electron distribution. <br> * Remains a fundamental teaching tool in chemistry. <br> * Allows scientists to easily show how electrons are shared within molecules. |
| Octet Rule | * Suggested that atoms tend to gain, lose, or share electrons to achieve a stable configuration of eight valence electrons (or two for hydrogen). | * Provided a predictive framework for understanding chemical reactivity. <br> * Helped explain the formation of many stable molecules. <br> * Created a set of easy-to-understand rules that helped in the understanding of chemical reactions. |
| Lewis Acid-Base Theory | * Redefined acids and bases as electron pair acceptors and donors, respectively. <br> * Expanded the scope of acid-base chemistry beyond proton transfer. | * Provided a more general and comprehensive understanding of acid-base reactions. <br> * Made it possible to understand reactions in non-aqueous solutions. <br> * Increased the scope of reactions that could be studied as acid/base reactions. |
Export to Sheets
Key Points:
- Paradigm Shift:
- Lewis’s work fundamentally changed how chemists understood chemical bonding.
- Visual Tools:
- Lewis dot structures provide a simple and effective way to represent molecular structure.
- Predictive Power:
- The octet rule, while not universally applicable, provides a useful predictive tool.
- Expanded Concepts:
- The Lewis acid-base theory increased the scope of acid-base chemistry.
Lewis Structures: His dot diagrams remain essential for chemists. Table
You’re right to emphasize the enduring importance of Lewis structures. They remain a fundamental tool in chemistry, and here’s a table illustrating their significance:
**Lewis Structures: Essential for Chemists
| Aspect | Description | Significance |
| Visual Representation of Bonding | * Lewis structures use dots to represent valence electrons and lines to represent shared electron pairs (covalent bonds). <br> * They provide a visual map of how atoms connect in a molecule. | * Simplify the understanding of chemical bonding and molecular structure. <br> * Allow chemists to quickly visualize electron distribution and bond formation. <br> * Are very useful tools when predicting molecular geometry. |
| Predicting Molecular Shape | * Lewis structures can be used in conjunction with VSEPR theory (Valence Shell Electron Pair Repulsion) to predict the three-dimensional shapes of molecules. <br> * Help to visualize lone pairs. | * Molecular shape is critical in understanding a molecule’s properties and reactivity. <br> * Provides an understanding of how molecules will interact with each other. <br> * Because molecular shape is so important in biological molecules, Lewis structures are also very important in the field of biochemistry. |
| Understanding Reactivity | * Lewis structures show the distribution of electrons, which helps predict how molecules will react. <br> * They highlight areas of electron density and potential reactive sites. | * Provides insights into chemical reactions and the mechanisms by which they occur. <br> * Help in understanding the chemical properties of different compounds. <br> * By allowing the prediction of chemical reactions, they have become a tool of chemical synthesis. |
| Fundamental teaching tool. | Lewis dot structures are often one of the first representational tools students learn when learning about molecular bonding. | The relatively easy concept allows even introductory chemistry students to visualize and begin to understand molecular bonding. |
Export to Sheets
Key Points:
- Simplicity and Power:
- Lewis structures provide a simple yet powerful way to represent molecular bonding.
- Visualizing the Invisible:
- They help chemists visualize the arrangement of electrons and atoms in molecules.
- Predictive Ability:
- They are valuable for predicting molecular shapes and reactivity.
- Educational Tool:
- Lewis structures remain a cornerstone of chemical education.
Lewis Acid-Base Theory: His acid-base theory is widely used, particularly in organic and inorganic chemistry. Table
Gilbert N. Lewis’s acid-base theory expanded the traditional understanding of acids and bases, making it particularly valuable in organic and inorganic chemistry. Here’s a table summarizing its key aspects:
Lewis Acid-Base Theory
| Aspect | Description | Significance |
| Electron Pair Concept | * Defines acids as electron pair acceptors and bases as electron pair donors. <br> * Focuses on the transfer or sharing of electron pairs rather than protons (as in Brønsted-Lowry theory). | * Broadens the definition of acids and bases beyond proton-containing substances. <br> * Applicable to a wider range of chemical reactions, including those in non-aqueous solutions and reactions involving metal ions. <br> * Creates a more inclusive definition of acid and base reactions. |
| Application to various reactions | Lewis’s definition applies to reactions where there is no proton transfer. Coordination chemistry and many organic mechanisms benefit from this view of acids and bases. | Many reactions, especially in organic and inorganic chemistry, can be explained with this model when the previous models were insufficient. |
| Formation of Coordinate Covalent Bonds | * Lewis acid-base reactions result in the formation of coordinate covalent bonds, where both electrons in the bond come from the base. | * Explain the formation of adducts and complexes. <br> * Helps understand the mechanisms of numerous chemical reactions. |
| Wide Applicability | Lewis’s definition of acids and bases increases the ability to describe many reactions. | This definition, due to its broader nature, has become very important in many fields of chemistry. |
Export to Sheets
Key Points:
- Expanded Scope:
- Lewis’s theory goes beyond the Brønsted-Lowry definition, encompassing reactions where proton transfer doesn’t occur.
- Electron Pair Focus:
- It emphasizes the role of electron pairs in acid-base reactions.
- Coordinate Covalent Bonds:
- It highlights the formation of coordinate covalent bonds in Lewis acid-base interactions.
- Versatility:
- This theory provides a powerful framework for understanding various types of reactions and significantly enhances our comprehension of numerous aspects of chemical interactions.
Lewis’s Influence on Physical Chemistry: His work on thermodynamics and other areas had a significant impact. Table
Gilbert N. Lewis’s impact on physical chemistry was broad and deep, extending beyond his well-known work on chemical bonding. He significantly shaped the field of thermodynamics and contributed to other areas as well. Here’s a table summarizing his influence:
Gilbert N. Lewis: Influence on Physical Chemistry
| Area of Influence | Description | Significance |
| Thermodynamics | * Developed and refined the concepts of fugacity and activity coefficients. <br> * Systematized thermodynamic data and applied thermodynamic principles to chemical systems. | * Enabled more accurate calculations of chemical equilibria and reaction rates, especially for real gases and non-ideal solutions. <br> * Provided a more rigorous and applicable foundation for chemical thermodynamics. <br> * his work increased the ability to model real-world chemical reactions. |
| Chemical Bonding | * Proposed the concept of the covalent bond and developed Lewis dot structures. | * Revolutionized the understanding of molecular structure and reactivity. <br> * Provided essential tools for visualizing and predicting chemical behavior. <br> * His work on bonding is the base of much of modern chemical theory. |
| Acid-Base Theory | * Developed the Lewis acid-base theory, expanding the definition of acids and bases. | * Broadened the scope of acid-base chemistry, particularly in organic and inorganic chemistry. <br> * Allowed for the study of reactions that previous acid base models could not describe. |
| Isotope Chemistry | * Contributed to the study of isotopes, including early work on heavy water (deuterium oxide). | * His work on isotopes helped to increase knowledge within the field of isotope chemistry. |
| Coined the term “photon” | He coined the term photon, which is still in use today. | His terminology is still used in modern physics and chemistry. |
Export to Sheets
Key Points:
- Broad Impact:
- Lewis’s contributions spanned multiple areas of physical chemistry.
- Emphasis on Real Systems:
- He focused on applying theoretical principles to real-world chemical systems.
- Lasting Legacy:
- His work continues to influence modern chemical research and education.
Lewis Thermodynamics: Made significant contributions to chemical thermodynamics, applying thermodynamic principles to chemical systems. Table
Gilbert N. Lewis’s contributions to chemical thermodynamics were substantial, providing a more rigorous and applicable framework for understanding chemical systems. Here’s a table summarizing his key contributions:
Gilbert N. Lewis: Contributions to Chemical Thermodynamics
| Contribution | Description | Significance |
| Fugacity | * Introduced the concept of fugacity, a measure of the escaping tendency of a substance in a real gas or liquid. <br> * Developed methods for calculating fugacity from experimental data. | * Extended the applicability of thermodynamic principles to real gases and solutions, which deviate from ideal behavior. <br> * Provided a more accurate description of chemical equilibrium and phase transitions. <br> * Allowed for more accurate thermodynamic calculations. |
| Activity Coefficients | * Developed the concept of activity coefficients to account for the non-ideal behavior of solutions. <br> * Introduced methods for determining activity coefficients. | * Enabled the accurate calculation of equilibrium constants and reaction rates in non-ideal solutions. <br> * Improved the understanding of the thermodynamic properties of concentrated solutions and electrolytes. <br> * Increased the applicability of thermodynamics to real-world solutions. |
| Systematization of Thermodynamic Data | * Compiled and organized vast amounts of thermodynamic data, making it more accessible and useful to chemists. <br> * Emphasized the importance of consistent and accurate thermodynamic measurements. | * Facilitated the application of thermodynamic principles to a wide range of chemical systems. <br> * Advanced the development of quantitative chemical thermodynamics. <br> * This vast cataloging and organization further advanced the field of physical chemistry. |
| Application to chemical equilibrium | Through his development of fugacity and activity coefficients, he significantly enhanced chemists’ ability to accurately predict chemical equilibria. | Allows the more accurate prediction of chemical reaction outcomes. |
Export to Sheets
Key Points:
- Real-World Application:
- Lewis’s work focused on applying thermodynamics to real chemical systems rather than idealized ones.
- Quantitative Approach:
- He emphasized the importance of quantitative measurements and calculations in thermodynamics.
- Fundamental Concepts:
- Fugacity and activity coefficients are now fundamental concepts in chemical thermodynamics.
- Increased Accuracy:
- By introducing these ideas and organizing thermodynamic data, chemical calculations became far more accurate.
Lewis “Photon” Term: Coined the term “photon” for the quantum of light (though his concept of the photon differed somewhat from the modern understanding). Table
It’s true that Gilbert N. Lewis coined the term “photon,” although it’s important to understand that his initial concept differed somewhat from the modern understanding of the photon. Here’s a breakdown:
Gilbert N. Lewis: “Photon” Term
| Feature | Description | Significance |
| Origin of the Term | * In a 1926 paper in “Nature,” Lewis proposed the term “photon” for the quantum of light. <br> * He intended it to describe a fundamental carrier of radiant energy. <br> * However, his model differed from the quantum mechanical model that later became established. | * Lewis’s term “photon” was eventually adopted by the scientific community to describe the particle of light, thus his lasting effect on the nomenclature of physics. <br> * While his original conception of the photon was not entirely correct, he is still credited with giving the photon its name. |
| His original theory | Lewis had proposed a theory where photons where indestructible, and had a structural quality, something that was later proven to be incorrect. | Although this theory was proven to be incorrect, his naming of the photon remained. |
| How the photon is understood today | The modern understanding of the photon describes it as an elementary particle and a quantum of electromagnetic radiation. | The modern understanding of the photon is ingrained within quantum mechanics. |
Export to Sheets
Key Points:
- Nomenclature Legacy:
- Lewis is credited with giving the photon its name, which is now universally used.
- Conceptual Differences:
- His original concept of the photon differed from the modern quantum mechanical understanding.
- Evolution of Understanding:
- The understanding of the photon has evolved significantly since Lewis’s time, along with the development of quantum mechanics.
I hope this helps to clarify the situation.
Lewis Heavy Water: The first isolate is a pure sample of Heavy Water. Table
You’re right to highlight Gilbert N. Lewis’s significant contribution to the isolation of pure heavy water. Here’s a table summarizing his achievement:
Gilbert N. Lewis: Isolation of Pure Heavy Water
| Aspect | Description | Significance |
| Deuterium and Heavy Water | * Deuterium is a heavier isotope of hydrogen (²H or D). <br> * Heavy water (D₂O) is water in which the “normal” hydrogen atoms (¹H) are replaced by deuterium atoms. | * The existence of deuterium and heavy water was of great scientific interest. <br> * They had potential applications in various scientific fields. |
| Lewis’s Achievement | * In 1933, Gilbert N. Lewis successfully isolated the first pure sample of heavy water. <br> * He used prolonged electrolysis of ordinary water to concentrate and obtain D₂O. | * This was a significant achievement in isotope chemistry and physical chemistry. <br> * It provided a pure source of heavy water for further research. <br> * Helped to validate Urey’s previous discovery of Deuterium. |
| Electrolysis Method | By using electrolysis to separate normal water, over very long time periods, the heavier water became more concentrated, until a pure sample was able to be taken. | Although slow, this process proved effective in gathering heavy water. |
| Significance of Heavy Water | * Heavy water is used as a moderator in nuclear reactors. <br> * It’s also used in isotopic labeling experiments in chemistry and biology. | * Lewis’s work enabled further studies and applications of heavy water. <br> * Advanced research in nuclear physics and other fields. |
Export to Sheets
Key Points:
- Isotopic Separation: Lewis’s work demonstrated a practical method for separating isotopes.
- Nuclear Relevance: Heavy water’s importance in nuclear technology highlighted the significance of Lewis’s achievement.
- Fundamental Research: His work laid the groundwork for further research into isotopes and their applications.
Linus Pauling; Quantum Chemistry

Pauling in the 1940s
(Wiki Image By Unknown author – Oregon State University [1], CC BY-SA 2.0, https://commons.wikimedia.org/w/index.php?curid=144490366)
Linus Pauling Quotes. Table
Let’s present a selection of quotes from Linus Pauling in a table format. His career spanned many decades and fields, so his quotes reflect his work in chemistry, molecular biology, peace activism, and general philosophy.
| Quote Number | Quote | Source | Context/Significance |
| 1 | “The best way to have a good idea is to have lots of ideas.” | Widely attributed | Reflects his prolific creativity and belief in generating many possibilities to find the best scientific solutions. |
| 2 | “Satisfaction of one’s curiosity is one of the greatest sources of happiness in life.” | Widely attributed | Expresses his deep personal motivation for scientific inquiry and the joy of discovery. |
| 3 | “Science is the search for truth – it is not a game in which one tries to beat his opponent, to do harm to others.” | Widely attributed | Highlights his view of science as a collaborative pursuit of knowledge, contrasting it with competitive or destructive endeavors. |
| 4 | “Facts are the air of scientists. Without them, you can never fly.” | Attributed | Emphasizes the crucial importance of empirical evidence and observation as the foundation for scientific theory and progress. |
| 5 | “I have always liked understanding things. It is a basic characteristic of scientists that they like to understand things.” | Interview | This simple statement reflects his fundamental curiosity and the driving force behind his scientific career. |
| 6 | “[Regarding chemical bonds] …the forces which hold atoms together in molecules and crystals…” (The Nature of the Chemical Bond, Introduction) | The Nature of the Chemical Bond (1939) | From the introduction to his landmark textbook, defining the central topic, he revolutionized by applying quantum mechanics. |
| 7 | “I believe that there is a greater power in the world than the evil power of military force, of nuclear bombs — there is the power of good, of morality, of humanitarianism.” | Statements related to his peace activism | This reflects his deep commitment to peace and nuclear disarmament, which earned him the Nobel Peace Prize. It also shows the ethical dimension of his work. |
| 8 | “Do unto others 20% better than you would expect them to do unto you, to correct for subjective error.” | Attributed; reflects his rational approach | A quirky and humorous reflection of his attempt to apply a rational, almost mathematical approach even to ethics, acknowledging human bias. |
| 9 | “Optimum nutrition is the medicine of tomorrow.” | Reflects his later (controversial) advocacy | Represents his strong belief in the power of vitamins (particularly Vitamin C) for health, a view not widely supported by mainstream medical science but one he passionately defended. |
| 10 | “Every aspect of the world is understandable on a continuous scale from atoms to galaxies.” | Lectures/Writings | Shows his belief in the unifying power of science to explain phenomena across different scales, linking chemistry to biology and the broader universe. |
Export to Sheets
Key Takeaways:
- Curiosity and Ideas: Pauling emphasized the importance of curiosity and generating numerous ideas.
- Truth and Evidence: He saw science as a search for truth based on factual evidence.
- Breadth: His quotes reflect his work across multiple fields – chemistry, biology, peace activism.
- Humanitarianism: He felt a strong sense of social responsibility, particularly regarding the dangers of nuclear weapons.
- Confidence (and Controversy): He held strong convictions, sometimes leading to controversy (like his later views on Vitamin C).
This table provides a glimpse into Linus Pauling’s mind, showcasing his scientific philosophy, humanitarian concerns, and wide-ranging intellect. It reflects a scientist who made fundamental discoveries and engaged deeply with the broader implications of science for humanity.
Linus Pauling YouTube Video
- Linus Pauling – Conversations with History by University of California Television (UCTV): 124,936 views (http://www.youtube.com/watch?v=WHzG3nTA27M)
- Linus Pauling Lecture: Valence and Molecular Structure Part 1 by Oregon State University: 65,383 views (http://www.youtube.com/watch?v=7tevU4Cu4XE)
- Remembering Linus Pauling by Oregon State University: 8,959 views (http://www.youtube.com/watch?v=SrVTV4jlpAM)
- Linus Pauling’s Groundbreaking Findings on Heart Health #hearthealth #linuspauling #heartattack by Peter Osborne: 2,926 views (http://www.youtube.com/watch?v=d9w41XLsSyc)
- Linus Pauling’s unscientific belief by FREE SCIENCE 365: 1,711 views (http://www.youtube.com/watch?v=iyZmh_w6ZrU)
Linus Pauling History

Linus Pauling with an inset of his Nobel Prize in 1955
(Wiki Image By Unknown author – The Big T (yearbook of California Institute of Technology), Public Domain, https://commons.wikimedia.org/w/index.php?curid=89967697)
Linus Pauling (1901-1994) was one of the most influential scientists of the 20th century. He made groundbreaking contributions to chemistry, physics, biology, and medicine. He was a brilliant theorist, a skilled experimentalist, and a passionate advocate for peace. He is the only person awarded two unshared Nobel Prizes (Chemistry in 1954 and Peace in 1962).
- Early Life and Education (1901-1925):
- Born in Portland, Oregon (1901): His father was a pharmacist, which may have sparked his early interest in chemistry.
- Early Interest in Science: Showed an early aptitude for science, collecting insects and minerals and conducting chemistry experiments.
- Oregon Agricultural College (OAC) (1917-1922): Studied chemical engineering at Oregon Agricultural College (now Oregon State University). He was a brilliant student but had to work to support himself and his family.
- California Institute of Technology (Caltech) (1922-1925): He pursued his Ph.D. in physical chemistry and mathematical physics at Caltech, under the supervision of Roscoe G. Dickinson. He was introduced to X-ray crystallography, a technique that would become central to his research.
- Doctoral Research: His doctoral research focused on determining the crystal structures of minerals using X-ray diffraction.
- Early Career and Quantum Chemistry (1925-1930s):
- Postdoctoral Studies in Europe (1926-1927): He spent time in Europe studying with leading physicists, including Arnold Sommerfeld, Niels Bohr, and Erwin Schrödinger. This exposed him to the newly developing field of quantum mechanics.
- Return to Caltech (1927): He joined the faculty at Caltech, where he would spend most of his career.
- Application of Quantum Mechanics to Chemistry: He became one of the first to apply quantum mechanics to explain the nature of the chemical bond. This was a revolutionary approach, as classical physics could not adequately explain how atoms bonded to form molecules.
- Valence Bond Theory: Developed valence bond theory describes chemical bonds as arising from overlapping atomic orbitals and sharing electrons. He used quantum mechanics to calculate the energies and properties of chemical bonds.
- Hybridization: Introduced the concept of orbital hybridization to explain the geometry of molecules (e.g., the tetrahedral arrangement of bonds in methane). This explains why carbon forms four bonds.
- Resonance: Developed the concept of resonance to describe molecules that cannot be adequately represented by a single Lewis structure (e.g., benzene). This explained the unusual stability of certain molecules.
- Electronegativity: Introduced the concept of electronegativity and developed the Pauling electronegativity scale, a measure of an atom’s ability to attract electrons in a chemical bond. This helped to predict the polarity of chemical bonds.
- The Nature of the Chemical Bond (1939): He published his landmark book, The Nature of the Chemical Bond and the Structure of Molecules and Crystals, which became one of the most influential chemistry texts of the 20th century. It summarized his work on chemical bonding and presented a unified framework for understanding molecular structure.
- Structural Biology and Medical Research (1930s-1960s):
- Focus on Biological Molecules: He became increasingly interested in applying his chemical bonding and molecular structure knowledge to biological problems.
- Protein Structure: He began studying the structure of proteins, using X-ray crystallography and his understanding of chemical bonding.
- Alpha Helix (1951): Proposed the alpha-helix structure, a fundamental structural element of proteins (a coiled structure).
- Beta Sheet (1951): Proposed the beta-sheet structure, another fundamental structural element of proteins (a folded sheet-like structure).
- These discoveries were breakthroughs in understanding protein structure and function.
- Sickle cell anemia is a molecular disease caused by a change in the structure of hemoglobin (the protein that carries oxygen in red blood cells). It was one of the first examples of a disease being linked to a specific molecular defect.
- DNA Structure: Made an early attempt to determine the structure of DNA, proposing an incorrect triple-helix model. While he was wrong, his work stimulated research in this area and influenced Watson and Crick.
- Activism and the Nobel Peace Prize (1950s-1960s):
- Concerns about Nuclear Weapons: Became deeply concerned about the dangers of nuclear weapons and nuclear testing after World War II.
- Advocate for Nuclear Disarmament: He became a prominent advocate for nuclear disarmament and international peace.
- Petition Against Nuclear Weapons Testing: Circulated a petition signed by thousands of scientists, calling for an end to nuclear weapons testing.
- Nobel Peace Prize (1962): He was awarded the Nobel Peace Prize for his efforts to ban nuclear weapons testing, making him the only person to win two unshared Nobel Prizes.
- Later Years and Vitamin C Controversy (1970s-1994):
- Vitamin C Advocacy: Became a strong advocate for high doses of vitamin C to prevent and treat diseases, including the common cold and cancer. This was highly controversial, and mainstream medical science did not support his views.
- Linus Pauling Institute of Science and Medicine: This institute promotes research on orthomolecular medicine (the use of vitamins and other natural substances to treat disease).
- Death (1994): He died in 1994 at 93.
- Legacy:
- One of the Greatest Scientists of the 20th Century: Widely considered one of the most influential scientists ever.
- Founder of Quantum Chemistry: His application of quantum mechanics to chemistry revolutionized the field.
- Fundamental Contributions to Molecular Biology: His work on protein structure was crucial for developing molecular biology.
- Two Nobel Prizes: He is the only person to win two unshared Nobel Prizes, reflecting the breadth and impact of his work.
- Influence on Multiple Fields: His contributions spanned chemistry, physics, biology, medicine, and peace activism.
- Scientific and Humanitarian Legacy: He left a lasting legacy as a brilliant scientist and a passionate advocate for peace and social responsibility.
Linus Pauling’s career was remarkable for its breadth, depth, and impact. He was a true polymath, making fundamental contributions to multiple scientific fields. His work on chemical bonding, protein structure, and molecular disease transformed our understanding of the molecular world, and his advocacy for peace made him a prominent figure in the 20th-century peace movement. While his later views on vitamin C remain controversial, his scientific achievements are undeniable, and his legacy as one of the most outstanding scientists of all time is secure.
Linus Pauling, Quantum Chemistry, and Legacy. Table
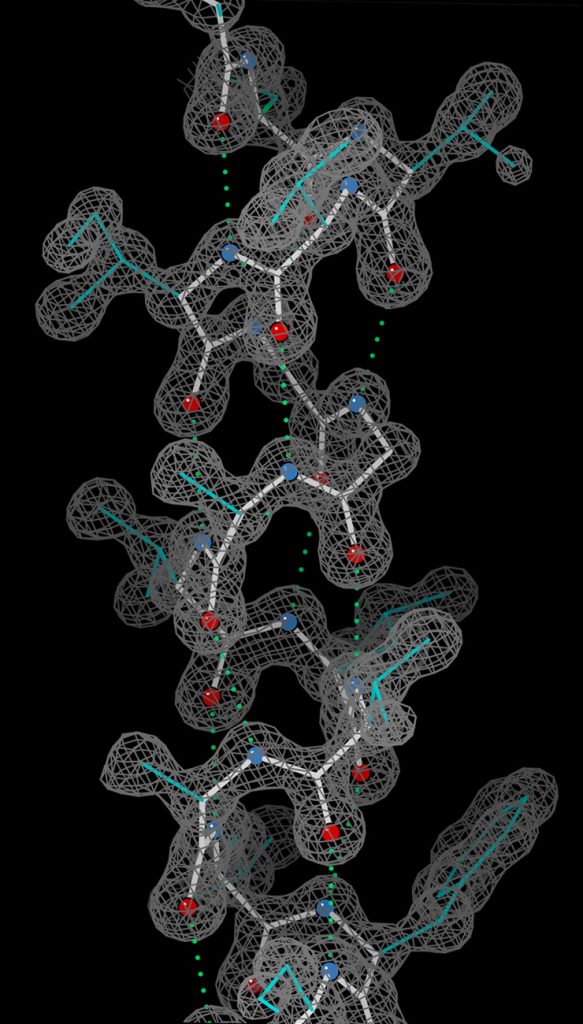
An alpha helix in ultra-high-resolution electron density contours, with O atoms in red, N atoms in blue, and hydrogen bonds as green dotted lines (PDB file 2NRL, 17–32)
(Wiki Image By Dcrjsr – Own work, CC BY 3.0, https://commons.wikimedia.org/w/index.php?curid=9131613)
Let’s present Linus Pauling’s (1901-1994) contributions in a structured table format, particularly his work in quantum chemistry and his broader legacy. This will highlight the key aspects of his multifaceted career.
| Aspect | Details | Significance |
| Early Life and Education | – Born in Portland, Oregon (1901). <br> – Early interest in science. <br> – B.S. in Chemical Engineering from Oregon Agricultural College (now Oregon State University) (1922). <br> – Ph.D. in Physical Chemistry and Mathematical Physics from Caltech (1925). | – A strong foundation in chemistry and physics, crucial for his later work in quantum chemistry. |
| Quantum Chemistry and Chemical Bonding | – Application of Quantum Mechanics to Chemistry: One of the first to apply the newly developed quantum mechanics to explain the nature of the chemical bond. <br> – Valence Bond Theory: The Developed valence bond theory describes chemical bonds as arising from overlapping atomic orbitals and sharing electrons. <br> – Hybridization: Introduced the concept of orbital hybridization to explain the geometry of molecules (e.g., the tetrahedral arrangement of bonds in methane). <br> – Resonance: Developed the concept of resonance to describe molecules that cannot be adequately represented by a single Lewis structure (e.g., benzene). <br> – Electronegativity: Introduced the concept of electronegativity and developed the Pauling electronegativity scale, a measure of an atom’s ability to attract electrons in a chemical bond. <br> – The Nature of the Chemical Bond (1939): He published his landmark book, one of the most influential chemistry texts of the 20th century. | – Revolutionized understanding of chemical bonding. Provided a theoretical framework for understanding why and how atoms combine to form molecules. <br> – His concepts (valence bond theory, hybridization, resonance, electronegativity) remain fundamental to chemical education and research. <br> – Bridged the gap between physics and chemistry. |
| Structural Chemistry and Biology | – X-ray Crystallography: X-ray crystallography determines the structures of crystals and molecules. <br> – Protein Structure: Determined the alpha-helix and beta-sheet structures, two fundamental structural elements of proteins. This was a breakthrough in understanding protein structure. <br> – Sickle Cell Anemia: Sickle cell anemia is a molecular disease caused by a change in the structure of hemoglobin. <br> – DNA Structure: He made an early attempt to determine the structure of DNA (though he proposed an incorrect triple-helix model). His work, however, influenced Watson and Crick. | – Pioneered the application of physical methods (X-ray crystallography) to determine the structures of complex molecules. <br> – Made fundamental contributions to structural biology. His work on protein structure was crucial for understanding the function of biological molecules. <br> – Helped to establish the field of molecular biology. |
| Activism and Peace Work | – Advocate for Nuclear Disarmament: Became a prominent advocate for nuclear disarmament and international peace after World War II. <br> – Petition Against Nuclear Weapons Testing: Circulated a petition signed by thousands of scientists, calling for an end to nuclear weapons testing. <br> – Nobel Peace Prize (1962): Awarded the Nobel Peace Prize for his efforts to ban nuclear weapons testing. | – Demonstrated his commitment to social responsibility and the ethical use of science. <br> – Became a leading voice in the movement against nuclear weapons. |
| Other Interests | – Vitamin C: Later in his career, he strongly advocated for high doses of vitamin C to prevent and treat diseases (this is controversial and not supported by mainstream medical science). | – He showed that he maintained an interest in many different areas. |
| Legacy | – One of the Greatest Chemists of All Time: Widely considered one of the most influential chemists of the 20th century. <br> – Two Nobel Prizes: The only person to win two unshared Nobel Prizes (Chemistry and Peace). <br> – Founder of Quantum Chemistry: His work laid the foundations for the field of quantum chemistry. <br> – Influence on Multiple Fields: His contributions spanned chemistry, physics, biology, and medicine. <br> – Scientific and Humanitarian Legacy: Left a lasting legacy as a brilliant scientist and passionate advocate for peace. | – His work continues to influence research in many areas of science. <br> – He is remembered as a scientific giant and a humanitarian. |
Export to Sheets
Key Takeaways:
- Interdisciplinary Approach: Pauling’s work exemplifies the power of a multidisciplinary approach, combining physics, chemistry, and biology.
- Theoretical and Experimental: He was a brilliant theorist and a skilled experimentalist.
- Impact on Multiple Fields: His contributions profoundly impacted multiple scientific disciplines.
- Social Responsibility: He was deeply concerned about scientists’ social responsibility and used his influence to advocate for peace.
- Controversial Figure: He was well respected, but some of his later work was questioned.
This table comprehensively overviews Linus Pauling’s life, scientific contributions, and legacy. It highlights his multifaceted career and profound impact on science and society. The format allows for a clear understanding of the progression of his work and the interconnectedness of his various achievements.
Pauling’s Application of Quantum Mechanics to Chemistry: Pauling was one of the first to apply the newly developed quantum mechanics to explain the nature of the chemical bond. Table. Pictures.
Okay, let’s break down Linus Pauling’s groundbreaking application of quantum mechanics to chemical bonding in a table format.
| Aspect of Quantum Mechanical Application | Details / Explanation |
| Context (Early 20th Century) | – G.N. Lewis had established the concept of the covalent bond as a shared electron pair but lacked a deep physical explanation of why or how they formed. <br> – Classical physics could not explain the stability or geometry of molecules. <br> – Quantum mechanics was emerging as a powerful new theory describing the behavior of electrons and atoms. |
| Pauling’s Pioneering Role | Pauling was one of the first and most influential scientists to bridge the gap between quantum mechanics (physics) and chemistry. He applied the complex mathematical framework of quantum mechanics to provide a theoretical understanding of the chemical bond. |
| Primary Theoretical Framework Used | Valence Bond (VB) Theory: Pauling significantly developed and popularized VB theory. This approach describes bond formation in terms of the overlap of atomic orbitals containing valence electrons. |
| Explaining the Covalent Bond | – Orbital Overlap: Pauling used quantum mechanics to show how atomic orbitals (regions where electrons exist) on adjacent atoms could overlap. <br> – Shared Electron Pair: He described the covalent bond as forming when two overlapping atomic orbitals are occupied by a pair of electrons with opposite spins. This shared pair is attracted to both nuclei, holding the atoms together. <br> – Bond Strength: Related bond strength to the degree of orbital overlap (greater overlap = stronger bond). |
| Explaining Molecular Geometry (Hybridization) | – Problem: The Simple overlap of standard atomic orbitals (s, p) could not explain the observed shapes of many molecules (e.g., the 109.5° angles in tetrahedral methane). <br> – Pauling’s Solution: Orbital Hybridization: He introduced the concept that atomic orbitals on the same atom could mathematically mix (hybridize) to form new hybrid orbitals (e.g., sp, sp², sp³) with specific shapes and directional properties suitable for forming bonds with the correct geometry. |
| Explaining Delocalized Bonding (Resonance) | – Problem: Some molecules (like benzene) couldn’t be represented accurately by a single Lewis structure. <br> – Pauling’s Solution: Resonance: He refined and popularized the concept of resonance, where the actual molecule is a quantum mechanical “hybrid” or average of multiple contributing structures. This explained the stability and bond lengths in molecules with delocalized electrons. |
| Explaining Bond Polarity (Electronegativity) | Pauling developed a quantitative electronegativity scale based on bond energies, providing a quantum mechanically informed way to understand the unequal sharing of electrons in polar covalent bonds (related to the concept of ionic character contributing to bond stability). |
| Major Publication | The Nature of the Chemical Bond (1939): His seminal book synthesized these ideas, presenting a comprehensive quantum mechanical view of chemical bonding and molecular structure. It became one of the most influential scientific books of the 20th century. |
| Significance | – Revolutionized the understanding of chemical bonding. <br> – Provided the first widely accepted theoretical basis for why atoms form molecules with specific shapes and properties. <br> – Unified diverse chemical phenomena (bond types, geometry, resonance) under a coherent quantum mechanical framework. <br> – Became fundamental to chemical education and research. |
Export to Sheets
In Summary:
Linus Pauling masterfully applied the principles of the new quantum mechanics to the long-standing chemical problem of the nature of the chemical bond. Through concepts like valence bond theory, orbital overlap, hybridization, resonance, and electronegativity, he provided a powerful and comprehensive theoretical framework that explained how atoms bond to form molecules, why molecules have specific shapes, and how their properties arise from their electronic structure, thereby revolutionizing chemistry.
Pauling Valence Bond Theory: The developed valence bond theory describes chemical bonds as arising from overlapping atomic orbitals and sharing electrons. Table.
Okay, let’s break down Linus Pauling’s Valence Bond Theory (VBT) into a table format, based on the provided description and its key features.
| Key Aspect of Valence Bond Theory | Description / Explanation |
| Theory Name | Valence Bond Theory (VBT) |
| Primary Developer/Popularizer | Linus Pauling (building on earlier work by Heitler, London, Slater) |
| Core Concept | Explain the formation of covalent chemical bonds between atoms. |
| Mechanism of Bond Formation | A covalent bond is formed when atomic orbitals (regions where valence electrons are likely found) from two different atoms overlap in space. |
| Nature of the Bond | The overlapping region contains a pair of electrons (usually one contributed by each atom) which are shared between the two bonded atoms. This shared electron pair attracts both nuclei, holding the atoms together. |
| Goal of Bonding | Atoms share electrons through orbital overlap primarily to achieve a more stable electron configuration, often resembling that of a noble gas (e.g., fulfilling the octet rule where applicable). |
| Theoretical Basis | VBT is a major application of quantum mechanics to chemistry, providing a more sophisticated and mathematically grounded description of bonding than earlier Lewis structures. |
| Key Concepts Developed/Explained by Pauling within VBT | – Hybridization: The concept of mixing atomic orbitals (s, p, d) to form new hybrid orbitals (e.g., sp, sp², sp³) with specific shapes and orientations suitable for explaining observed molecular geometries (like the tetrahedral shape of methane). <br> – Resonance: A way to describe molecules whose bonding cannot be accurately represented by a single Lewis structure. The actual structure is considered a hybrid or average of multiple contributing resonance structures. |
| Impact/Significance | – Revolutionized the understanding of chemical bonding. <br> – Provided a powerful theoretical framework for explaining molecular structure, geometry, and properties. <br> – Became a cornerstone of chemical education for decades. <br> – Successfully explained the bonding in a vast number of molecules. |
Export to Sheets
In Summary:
Linus Pauling’s Valence Bond Theory explains covalent bonding as the result of overlapping atomic orbitals containing a shared pair of electrons. This approach, rooted in quantum mechanics and incorporating concepts like hybridization and resonance, provided chemists with a powerful tool to understand and predict the structure and properties of molecules, fundamentally changing the field.
Pauling Hybridization: Introduced the concept of orbital hybridization to explain the geometry of molecules (e.g., the tetrahedral arrangement of bonds in methane). Table.
Okay, let’s break down Linus Pauling’s concept of orbital hybridization into a table format, explaining its purpose, mechanism, and significance.
| Aspect of Hybridization | Description / Explanation |
| Concept Name | Orbital Hybridization |
| Introduced By | Linus Pauling (as a key component of his Valence Bond Theory) |
| Purpose | To explain the observed geometries (shapes and bond angles) of molecules which could not be rationalized by the direct overlap of simple, unmixed atomic orbitals (s, p, d). It reconciles the predicted shapes based on electron pairs (VSEPR theory) with the shapes of atomic orbitals used in bonding. |
| Core Idea | Atomic orbitals within the same atom (specifically, valence orbitals like s and p) can mathematically mix or combine to form a new set of hybrid orbitals. These hybrid orbitals have different shapes, energies, and spatial orientations compared to the original atomic orbitals. |
| Characteristics of Hybrid Orbitals | – The number of hybrid orbitals formed equals the number of atomic orbitals that were mixed. <br> – The hybrid orbitals formed are generally equivalent to each other in shape and energy (e.g., the four sp³ orbitals are identical). <br> – They arrange themselves in space to minimize electron-pair repulsion, resulting in specific geometries that match experimentally observed shapes (e.g., tetrahedral, trigonal planar, linear). |
| Mechanism | It is a mathematical model used within the Valence Bond Theory. It describes how atomic orbitals could combine to produce orbitals with the correct orientation to match the experimentally observed bond angles and molecular shapes. It’s not necessarily a physical “mixing” process but a way to construct orbitals suitable for bonding. |
| Example: Methane (CH₄) | – Observation: Methane is tetrahedral with bond angles of 109.5°. <br> – Problem: Carbon’s ground state electron configuration (2s², 2p²) doesn’t readily explain four equivalent bonds at 109.5° angles using standard s and p orbitals (p orbitals are at 90° to each other). <br> – Hybridization Solution: Pauling proposed that carbon’s one 2s and three 2p valence orbitals hybridize to form four equivalent sp³ hybrid orbitals. These sp³ orbitals point toward the corners of a tetrahedron (109.5° angles), perfectly explaining methane’s geometry. Each sp³ orbital then overlaps with a hydrogen 1s orbital. |
| Other Common Types | – sp² Hybridization: (Mixing one s + two p orbitals) -> Three sp² orbitals arranged in a trigonal planar geometry (120° angles). Examples: Ethene (C₂H₄), Boron trifluoride (BF₃). <br> – sp Hybridization: (Mixing one s + one p orbital) -> Two sp orbitals arranged in a linear geometry (180° angles). Examples: Ethyne (C₂H₂), Beryllium chloride (BeCl₂). |
| Significance | – Crucial Extension of Valence Bond Theory: Allowed VBT to accurately predict and explain the observed three-dimensional shapes of molecules. <br> – Fundamental Concept: Became a fundamental concept in understanding chemical bonding and molecular structure, particularly in organic chemistry. <br> – Widely Taught: Remains an integral part of chemical education for explaining molecular geometry. |
Export to Sheets
In Summary:
Orbital hybridization, as introduced by Linus Pauling, is a theoretical concept within Valence Bond Theory that explains how atomic orbitals mix to form new hybrid orbitals. These hybrid orbitals have the correct shapes and spatial orientations to account for the experimentally observed geometries of molecules, providing a powerful tool for understanding and predicting molecular structure.
Pauling Resonance: Developed the concept of resonance to describe molecules that cannot be adequately represented by a single Lewis structure (e.g., benzene). Table.
Okay, let’s break down Linus Pauling’s concept of Resonance into a table format, explaining its purpose, core idea, characteristics, and significance.
| Aspect of Resonance | Description / Explanation |
| Concept Name | Resonance (also sometimes called Mesomerism) |
| Developed/Popularized By | Linus Pauling (significantly developed and applied the concept within Valence Bond Theory) |
| Problem Addressed | Certain molecules or ions (e.g., benzene, ozone, carbonate ion, nitrate ion) cannot be accurately represented by a single valid Lewis structure. Multiple valid Lewis structures can be drawn, differing only in the placement of electrons (typically pi electrons or lone pairs). |
| Core Idea | The actual structure of the molecule/ion is not any one of the individual contributing Lewis structures (called resonance structures or canonical forms). Instead, the true structure is a resonance hybrid, which is an average or blend of all the valid contributing resonance structures. The contributing structures themselves do not exist individually. |
| Key Characteristics | – Delocalization of Electrons: Resonance describes the delocalization of electrons (usually pi electrons or lone pairs) over three or more atoms. These electrons are not confined between just two atoms or on a single atom but are spread out. <br> – Increased Stability: Molecules or ions exhibiting resonance are generally more stable (have lower energy) than would be expected from any single contributing resonance structure. This extra stability is called “resonance energy.” <br> – Intermediate Bond Lengths/Orders: Bonds involved in resonance often have lengths and strengths that are intermediate between typical single, double, or triple bonds (e.g., all C-C bonds in benzene are identical and intermediate between single and double). <br> – Contributing Structures: The individual resonance structures are theoretical constructs. The more stable a contributing structure (e.g., fewer formal charges, full octets), the more it resembles the actual resonance hybrid. |
| Representation | Resonance is indicated by drawing the contributing Lewis structures separated by a double-headed arrow (↔). This arrow specifically denotes resonance and should not be confused with equilibrium arrows (⇌). |
| Example: Benzene (C₆H₆) | – Benzene’s actual structure is a planar hexagon with all six carbon-carbon bonds being identical in length and strength. <br> – No single Lewis structure with alternating single and double bonds can accurately represent this. <br> – Resonance Explanation: Benzene is described as a resonance hybrid of primarily two contributing structures (with double bonds shifted). The actual structure has the pi electrons delocalized evenly around the entire ring, leading to the observed identical bond lengths and significant stability (aromaticity). |
| Significance | – Provided a crucial way (within Valence Bond Theory) to describe bonding in molecules where electrons are delocalized. <br> – Essential for understanding the structure, stability, and reactivity of many important molecules, particularly aromatic compounds (like benzene) and conjugated systems. <br> – It remains a fundamental concept in organic chemistry and is widely used to explain molecular properties. |
Export to Sheets
In Summary:
Resonance, as developed by Linus Pauling, is a theoretical concept used when a single Lewis structure fails to adequately describe the bonding in a molecule or ion. It posits that the true structure is a hybrid of multiple contributing structures, resulting in electron delocalization and increased stability. This concept is vital for accurately understanding the structure and behavior of many important chemical species.
Pauling Electronegativity: Introduced the concept of electronegativity and developed the Pauling electronegativity scale, a measure of an atom’s ability to attract electrons in a chemical bond. Table.
Okay, let’s break down Linus Pauling’s concept of Electronegativity and his scale into a table format.
| Aspect of Electronegativity | Description / Explanation |
| Concept Name | Electronegativity (symbolized often by the Greek letter chi, χ) |
| Introduced/Developed By | Linus Pauling (popularized the concept and developed the first widely used quantitative scale) |
| Definition | A chemical property that describes the relative ability of an atom to attract shared electrons (or electron density) towards itself when that atom is part of a chemical bond. |
| Core Idea | In a covalent bond between two different atoms (e.g., H-Cl), the electrons involved in the bond are usually not shared equally. The atom with the higher electronegativity exerts a stronger pull on the shared electron pair. |
| Consequence of Unequal Sharing | Leads to the formation of polar covalent bonds. The more electronegative atom acquires a partial negative charge (δ−), and the less electronegative atom acquires a partial positive charge (δ+). This creates a bond dipole moment. |
| Relation to Bond Type | The difference in electronegativity (Δχ) between two bonded atoms can be used to predict the general type of bond: <br> – Large Δχ: Bond is primarily ionic (electrons essentially transferred). <br> – Moderate Δχ: Bond is polar covalent (electrons shared unequally). <br> – Small or Zero Δχ: Bond is nonpolar covalent (electrons shared relatively equally). |
| Pauling Electronegativity Scale | – Development: The first quantitative scale was developed by Pauling in the 1930s. <br> – Basis: Derived from bond energy data. Pauling observed that the energy of a bond between two different atoms (A-B) was typically greater than the average of the energies of the A-A and B-B bonds. He attributed this extra energy to the ionic character of the bond, which is related to the electronegativity difference. <br> – Values: An arbitrary, relative scale. Fluorine (the most electronegative element) was assigned a value close to 4.0 (3.98), and values for other elements were calculated relative to this. <br> – Units: The Pauling scale is dimensionless. |
| Periodic Trends | Electronegativity generally: <br> – Increases across a period (from left to right) on the periodic table (due to increasing effective nuclear charge). <br> – Decreases down a group (from top to bottom) on the periodic table (due to increasing atomic size and shielding). <br> – Highest: Fluorine (F). <br> – Lowest: Francium (Fr) and Cesium (Cs). |
| Significance | – Provides a crucial tool for predicting the polarity of chemical bonds and, consequently, the polarity of entire molecules. <br> – Helps explain many physical properties of substances (e.g., melting point, boiling point, solubility) which depend on bond polarity and intermolecular forces. <br> – Fundamental concept in understanding chemical reactivity and reaction mechanisms. <br> – Widely used in all areas of chemistry, from introductory education to advanced research. |
Export to Sheets
In Summary:
Linus Pauling introduced electronegativity as a measure of an atom’s electron-attracting ability within a bond. His scale, based on bond energies, provided the first quantitative way to compare this property across different elements. The concept is fundamental for predicting bond types, understanding molecular polarity, and explaining a wide range of chemical properties and behaviors.
Pauling X-ray Crystallography: X-ray crystallography determines the structures of crystals and molecules. Table.
Okay, let’s create a table detailing Linus Pauling’s use and application of X-ray Crystallography in his work. While he didn’t invent the technique, he was a master at applying it and interpreting its results to understand molecular structure and chemical bonding.
| Aspect of Pauling & X-ray Crystallography | Details / Explanation |
| Technique Name | X-ray Crystallography (also X-ray Diffraction) |
| Basic Principle | An experimental technique where X-rays are beamed at a crystal. The regular arrangement of atoms within the crystal diffracts (scatters) the X-rays in a specific pattern. Analyzing this diffraction pattern mathematically allows scientists to deduce the precise three-dimensional arrangement of atoms in the molecule, including bond lengths and bond angles. |
| Pauling’s Role | Pauling was not the inventor of the technique, but he was an early adopter and highly skilled practitioner and interpreter. He recognized its immense power for determining the exact structure of molecules and crystals, which was crucial for validating and refining his theories of chemical bonding. |
| Early Applications (Minerals & Simple Compounds) | In his early career (1920s-1930s), Pauling used X-ray crystallography extensively to determine the structures of various minerals and simple inorganic/organic crystals. |
| Link to Bonding Theories | The precise data on bond lengths and angles obtained from these early X-ray studies provided crucial experimental evidence for Pauling’s developing theories on the nature of the chemical bond, including concepts like ionic radii, covalent radii, resonance, and electronegativity. |
| Application to Biological Molecules (Proteins) | Pauling later turned his attention to the much more complex structures of biological macromolecules, particularly proteins. |
| Prediction of Protein Secondary Structures (Alpha-Helix & Beta-Sheet) | While direct X-ray crystallography of large proteins was still very challenging, Pauling used his deep knowledge of chemical bonding principles (derived partly from X-ray studies of simpler amino acids and peptides), combined with meticulous model building, to predict stable, regular structures for polypeptide chains. This led to his proposal of the alpha-helix and beta-sheet structures in 1951 (with Robert Corey). |
| Confirmation by Others | Pauling’s predicted alpha-helix and beta-sheet structures were later confirmed as common structural motifs in globular proteins through X-ray crystallography work carried out by other researchers (like Max Perutz and John Kendrew). |
| DNA Structure Attempt | Pauling also used model building based on chemical principles and available (though limited) X-ray data to propose a structure for DNA. However, his proposed triple-helix structure was incorrect, famously losing the race to Watson and Crick who proposed the correct double helix. |
| Significance for Pauling’s Work | – Provided essential experimental data that grounded and validated his revolutionary theories of chemical bonding. <br> – Enabled his groundbreaking theoretical predictions of protein secondary structures. <br> – Demonstrated his ability to integrate experimental data with theoretical principles to understand molecular structure at all levels of complexity. |
Export to Sheets
In Summary:
X-ray crystallography was a vital experimental tool for Linus Pauling. He used it masterfully to determine the structures of simpler substances, which provided the empirical foundation for his theories on chemical bonding. He then applied the principles derived from these studies, combined with model building, to make groundbreaking predictions about the structures of complex biological molecules like proteins, profoundly influencing the development of structural biology.
Pauling Protein Structure: Determined the alpha-helix and beta-sheet structures, two fundamental structural elements of proteins. This was a breakthrough in understanding protein structure. Table.
Okay, let’s create a table detailing Linus Pauling’s work (in collaboration with Robert Corey and others) on determining the alpha-helix and beta-sheet structures of proteins.
| Aspect of Protein Structure Discovery | Details / Explanation |
| Key Discovery | Alpha-Helix (α-helix) and Beta-Sheet (β-sheet) Structures |
| Primary Researchers | Linus Pauling and Robert Corey (and colleagues at Caltech) |
| Year Proposed | 1951 (Published in a series of papers) |
| Problem Addressed | Understanding how the long polypeptide chains of proteins fold into regular, stable three-dimensional structures. Determining these structures (protein secondary structure) was crucial for understanding protein function. |
| Methodology Used | – Analysis of the Peptide Bond: Based on X-ray diffraction data from simpler molecules (amino acids, small peptides), Pauling and Corey established the precise geometry of the peptide bond connecting amino acids, recognizing its planar and rigid nature. <br> – Chemical Principles: Applied fundamental principles of chemical bonding, including known bond lengths, bond angles, steric hindrance (atoms not bumping into each other), and the importance of hydrogen bonding for stability. <br> – Meticulous Model Building: Used physical, scale models to systematically explore possible conformations (folding patterns) of polypeptide chains that were chemically plausible and maximized stabilizing hydrogen bonds between backbone atoms. |
| The Alpha-Helix (α-helix) | – Structure: A right-handed coiled or helical structure. <br> – Hydrogen Bonding: Stabilized by hydrogen bonds between the C=O group of one amino acid and the N-H group of the amino acid four residues further down the chain. These hydrogen bonds run nearly parallel to the axis of the helix. <br> – Prediction: Proposed as a highly stable and common way for a polypeptide chain to fold, satisfying all chemical constraints. |
| The Beta-Sheet (β-sheet) | – Structure: Composed of laterally connected strands (beta strands) of polypeptide chain, forming a generally twisted, pleated sheet. <br> – Hydrogen Bonding: Stabilized by hydrogen bonds between C=O and N-H groups on adjacent strands. Strands can run in the same direction (parallel sheet) or in opposite directions (antiparallel sheet). <br> – Prediction: Proposed as another stable, regular structure utilizing backbone hydrogen bonding. |
| Confirmation | Pauling and Corey’s proposed α-helix and β-sheet structures were later confirmed experimentally by X-ray crystallographers (e.g., Max Perutz, John Kendrew) as common and fundamental structural elements found within many complex globular proteins. |
| Significance | – Breakthrough in Structural Biology: Provided the first detailed, chemically plausible models for protein secondary structure. <br> – Foundation for Protein Folding: Laid the foundation for understanding how proteins achieve their complex three-dimensional shapes, which dictates their biological function. <br> – Impact on Biochemistry & Medicine: Crucial for understanding enzyme mechanisms, protein interactions, antibody structure, and the molecular basis of diseases involving protein misfolding. <br> – Demonstrated Pauling’s Method: Showcased his powerful approach of combining fundamental chemical principles with model building to predict complex biological structures. |
Export to Sheets
In Summary:
Linus Pauling’s prediction of the alpha-helix and beta-sheet structures, based on fundamental chemical principles and model building, was a landmark achievement in structural biology. These discoveries provided the crucial framework for understanding protein secondary structure and paved the way for determining the complete three-dimensional structures of proteins, revolutionizing biochemistry and molecular biology.
Pauling Sickle Cell Anemia: Showed that sickle cell anemia is a molecular disease caused by a change in the structure of hemoglobin. Table.
Okay, let’s break down Linus Pauling’s pivotal work on sickle cell anemia into a table format. This research demonstrated for the first time that a disease could be traced to a specific molecular abnormality.
| Aspect of Sickle Cell Anemia Discovery | Details / Explanation |
| Disease Studied | Sickle Cell Anemia: A hereditary blood disorder causing red blood cells to become rigid and sickle-shaped under low oxygen conditions. This leads to blocked blood flow, pain, organ damage, and anemia. |
| Prevailing View (Before Pauling) | The underlying cause of the red blood cell sickling was unknown. It was recognized as a disease affecting red blood cells, likely inherited, but the specific molecular defect was not identified. |
| Pauling’s Hypothesis (Mid-1940s) | Pauling hypothesized that sickle cell anemia might be caused by a subtle abnormality in the hemoglobin molecule itself, the protein responsible for carrying oxygen. He suspected a genetic difference led to a structurally different protein. |
| Key Researchers | Linus Pauling, Harvey Itano, S. J. Singer, and Ibert C. Wells (at Caltech). |
| Experimental Technique | Electrophoresis: A technique used to separate molecules based on their net electrical charge. Hemoglobin samples from different individuals were placed in an electric field. |
| Key Observation (1949) | Hemoglobin from individuals with sickle cell anemia (HbS) migrated differently in the electric field compared to normal hemoglobin (HbA). Hemoglobin from individuals with the sickle cell trait (carriers, genotype AS) showed both patterns (a mix of HbA and HbS). |
| Conclusion | The difference in electrophoretic migration proved that HbS and HbA had different electrical charges, indicating a difference in their molecular structure. This difference was linked directly to the presence or absence of the disease state. |
| Publication | “Sickle Cell Anemia, a Molecular Disease” was published in the journal Science in 1949. |
| Significance | – Established the concept of “Molecular Disease”: This was the first time a specific disease was definitively linked to an alteration in a single protein molecule. <br> – Foundation for Molecular Medicine: Revolutionized the understanding of genetic diseases, showing they could result from specific changes in protein structure. <br> – Understanding Protein Structure-Function: Provided powerful evidence for the crucial link between a protein’s precise molecular structure and its biological function (and how defects cause disease). <br> – Impact on Genetics: Supported the idea that genes dictate the structure of proteins. (The specific amino acid change in HbS was identified later by Vernon Ingram). |
Export to Sheets
In Summary:
Linus Pauling and his colleagues used electrophoresis to demonstrate that individuals with sickle cell anemia have a structurally different form of hemoglobin (HbS) compared to normal hemoglobin (HbA). This groundbreaking work established sickle cell anemia as the first identified “molecular disease,” proving that a specific alteration in a protein molecule could be the direct cause of a disease, thereby laying the foundation for molecular medicine.
Pauling DNA Structure: He made an early attempt to determine the structure of DNA (though he proposed an incorrect triple-helix model). His work, however, influenced Watson and Crick. Table.
Okay, let’s create a table detailing Linus Pauling’s attempt to determine the structure of DNA, his proposed model, its flaws, and its influence on the eventual discovery by Watson and Crick.
| Aspect of Pauling’s DNA Structure Attempt | Details / Explanation |
| Context | Mid-20th century race to determine the 3D structure of DNA, the molecule carrying genetic information. Key players included Pauling (Caltech), Watson & Crick (Cambridge), and Franklin & Wilkins (King’s College London). |
| Pauling’s Approach | Applied methods similar to his successful work on protein secondary structures: <br> – Heavy reliance on model building. <br> – Use of known chemical principles (bond lengths, angles). <br> – Interpretation of available X-ray diffraction data (though he reportedly had limited access to the best images, particularly Rosalind Franklin’s “Photo 51”). |
| Proposed Model (Published early 1953) | Proposed a Triple Helix structure. |
| Key Features of Pauling’s Model | – Three intertwined helical strands. <br> – The sugar-phosphate backbones were located tightly packed on the inside of the helix. <br> – The nitrogenous bases (A, T, C, G) were pointing outwards from the central axis. |
| Key Flaws in Pauling’s Model | – Phosphate Groups: Placing the negatively charged phosphate groups on the inside was chemically implausible. The strong negative charges would repel each other intensely, making the structure highly unstable. He also incorrectly depicted them as non-ionized (protonated). <br> – Bases Outward: This arrangement didn’t easily explain Chargaff’s rules (A pairs with T, C pairs with G) or suggest a mechanism for DNA replication or information storage. <br> – Based on Limited/Flawed Data: Pauling’s model did not adequately fit the X-ray diffraction data available (especially Franklin’s higher-quality data showing the phosphates clearly on the outside) and reportedly used incorrect density/water content values. |
| Influence on Watson and Crick | – Competition/Urgency: Pauling’s publication spurred Watson and Crick to accelerate their own model-building efforts, knowing a world-renowned chemist was close. <br> – Learning from Errors: Seeing the obvious flaws in Pauling’s model (especially the inward-facing phosphates) reinforced Watson and Crick’s conviction that the phosphates must be on the outside and helped them avoid similar mistakes. <br> – Validated Model Building: Pauling’s use of model building, despite the incorrect result for DNA, validated the general approach that Watson and Crick were also pursuing. |
| Outcome | Shortly after Pauling published his incorrect triple helix model, Watson and Crick published their correct double helix model (April 1953), which successfully incorporated the experimental data (X-ray diffraction, Chargaff’s rules) and chemical constraints. |
| Significance of Pauling’s Attempt | – Illustrates the intense scientific competition of the era. <br> – This shows that even brilliant scientists can make errors, often due to incomplete or misinterpreted data. <br> – Demonstrates the importance of accurate experimental data (like Franklin’s) in guiding model building. <br> – Served as an indirect catalyst for the final correct solution by increasing the urgency for Watson and Crick. |
Export to Sheets
In Summary:
Linus Pauling, a giant in structural chemistry, made a significant but ultimately incorrect attempt to determine the structure of DNA, proposing a triple helix with phosphates on the inside. While flawed, his model spurred on the competition, and the recognition of its errors helped guide James Watson and Francis Crick towards their successful double helix model. It stands as a fascinating episode in the history of science, highlighting the interplay of theory, experiment, competition, and even error in scientific discovery.
Dorothy Crowfoot Hodgkin; X-ray Crystallography

Dorothy Hodgkin, as Chancellor of the University of Bristol
(Wiki Image By University of Bristol – https://www.flickr.com/photos/bristoluniversity/45949493842/, CC BY-SA 2.0, https://commons.wikimedia.org/w/index.php?curid=94832670)
Dorothy Crowfoot Hodgkin Quotes. Table
Let’s organize a selection of quotes from Dorothy Crowfoot Hodgkin in a table format. Known for her meticulous work and perhaps less for her flamboyant public statements, her quotes often reflect her dedication, perseverance, and deep engagement with the science of crystallography.
| Quote Number | Quote | Source | Context/Significance |
| 1 | “I was captured for life by chemistry and by crystals.” | Widely attributed; reflecting on her early passion | Encapsulates her Lifelong Passion: This quote perfectly summarizes her early fascination and enduring dedication to the study of chemistry and the intricate structures revealed by crystals through X-ray diffraction. |
| 2 | “It has taken us nearly 35 years to get here.” | Representative statement regarding the solving of the insulin structure | Highlights Perseverance: Reflects the incredible timescale and dedication required to solve the complex structure of insulin, a project she began in the 1930s and finally completed in 1969. Demonstrates her extraordinary persistence. |
| 3 | “The understanding of the spatial arrangements of atoms in molecules is of fundamental importance to chemistry.” | Reflects the core principle of her work | Explain the Goal of Her Work: The central goal of X-ray crystallography is to articulate the fundamental importance of knowing the precise three-dimensional structure of molecules to understand their function and properties. |
| 4 | “I don’t feel that there are any particular difficulties about being a woman in science… except for the interruptions.” | Attributed, possibly somewhat understated | On Being a Woman in Science: This essay reflects her pragmatic approach and focuses on the scientific work while subtly acknowledging the societal/domestic interruptions women often faced (she raised three children while pursuing groundbreaking research). |
| 5 | “One has to be very persistent, I suppose.” | Often cited regarding her work, particularly on insulin | Understated Determination: A characteristically understated comment that speaks volumes about the immense patience, meticulousness, and unwavering determination required for her complex crystallographic work. |
| 6 | “[On receiving the Nobel Prize] I hope this award will encourage other women to take up science.” | Paraphrased from statements made around her Nobel Prize (1964) | Role Model: She demonstrates her awareness of her position as a role model and her desire to encourage more women to pursue careers in scientific fields. |
| 7 | “It is essential that we understand the implications of our scientific discoveries for peace and welfare.” | Representative of her views expressed through Pugwash involvement | Social Responsibility of Science: This reflects her later life’s commitment to peace activism and the belief that scientists are responsible for considering the societal impact of their work. |
Export to Sheets
Key Takeaways:
- Dedication to Crystallography: Her passion for understanding structures through crystals is evident.
- Perseverance: Her quotes reflect the immense patience and persistence required for her complex, long-term projects.
- Understated Manner: She often expressed profound ideas or immense effort in a characteristically understated way.
- Social Consciousness: Beyond her science, she was deeply concerned with peace and the societal implications of scientific work.
- Female Pioneer: Her statements subtly acknowledge her role as a woman in a male-dominated field.
This table provides a structured overview of key statements reflecting Dorothy Crowfoot Hodgkin’s scientific focus, dedication, and broader views. It captures the essence of a brilliant scientist renowned for her meticulous work, groundbreaking discoveries, and quiet determination.
Dorothy Crowfoot Hodgkin YouTube Video
- The exceptional life of Dorothy Crowfoot Hodgkin | The Royal Society: 27,773 views (http://www.youtube.com/watch?v=PraybTdD_9I)
- The Dorothy Hodgkin Fellowship by The Royal Society: 6,828 views (http://www.youtube.com/watch?v=xDsl0IHHiR4)
- Minerals, molecules & the Nobel Prize: Dorothy Crowfoot Hodgkin at the University Museum by Oxford University Museum of Natural History: 1,112 views (http://www.youtube.com/watch?v=X6vJMUNwRq4)
- The Dorothy Hodgkin Memorial Lecture 2012 by Somerville College, Oxford: 2,037 views (http://www.youtube.com/watch?v=KXjaNQCH_ls)
- Dorothy Hodgkin Lecture | Diabetes UK Professional Conference 2018 | Diabetes UK by Diabetes UK: 368 views (http://www.youtube.com/watch?v=SfA1BCOMJTo)
Dorothy Crowfoot Hodgkin’s History

A three-dimensional contour map of the electron density of penicillin derived from x-ray diffraction. The points of highest density show the positions of individual atoms in the penicillin. Hodgkin used this device to deduce the structure.
(Wiki Image By Museum of the History of Science, University of Oxford – Museum of the History of Science, University of Oxford, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=29019144)

Hodgkin built a molecular model of penicillin using the electron density contour maps behind the model.
(Wiki Image By Science Museum London / Science and Society Picture Library – Molecular model of penicillin by Dorothy M Crowfoot Hodgkin, England, 1945Uploaded by Mrjohncummings, CC BY-SA 2.0, https://commons.wikimedia.org/w/index.php?curid=28024341)
Dorothy Crowfoot Hodgkin (1910-1994) was a British chemist who pioneered the technique of X-ray crystallography to determine the three-dimensional structures of complex biological molecules. Her work profoundly impacted chemistry, biology, and medicine, and she was awarded the Nobel Prize in Chemistry in 1964. She is considered one of the most important scientists of the 20th century and a key figure in the development of structural biology.
- Early Life and Education (1910-1932):
- Born in Cairo, Egypt (1910), her father was an archaeologist and colonial administrator, and her mother was an expert in ancient textiles. She spent her early life in Egypt, England, and Sudan.
- Early Interest in Science: Developed an early interest in science, particularly chemistry and crystals, inspired by a book on X-ray crystallography.
- Education at Somerville College, Oxford (1928-1932): Studied chemistry at Somerville College, Oxford, one of the few colleges for women at the time. She was introduced to X-ray crystallography during her undergraduate studies.
- Became interested in X-rays: She became curious after a family friend gave her a lecture.
- Doctoral and Early Postdoctoral Research (1932-1940s):
- Doctoral Studies at Cambridge (1932-1934): Pursued her doctoral research at the University of Cambridge under the supervision of J.D. Bernal, a pioneer in applying X-ray crystallography to biological molecules. She worked on the structure of sterols.
- Return to Oxford (1934): She returned to Somerville College, Oxford, as a tutor and fellow and established her X-ray crystallography laboratory, which was unusual for a woman at the time.
- Early Work on Protein Crystallography: She began working on the structure of insulin, a project that would occupy her for over thirty years. This was an extremely ambitious undertaking, as proteins were far more complex than any molecules analyzed by X-ray crystallography before.
- Married Thomas Hodgkin
- Penicillin Structure (1942-1945): During World War II, she worked on the structure of penicillin, a crucial antibiotic. Her determination of the structure of penicillin was a major breakthrough, enabling its mass production and saving countless lives. This collaborative effort involved scientists in both the UK and the US.
- Major Structure Determinations (1940s-1960s):
- Vitamin B12 Structure (1948-1956): After years of painstaking work, Hodgkin and her team determined the structure of vitamin B12, a very complex molecule essential for red blood cell formation and nerve function. This was a monumental achievement in X-ray crystallography, demonstrating the technique’s power to solve incredibly complex structures. The molecule was far larger and more complex than anything previously solved.
- Nobel Prize in Chemistry (1964): She was awarded the Nobel Prize in Chemistry “for her determinations by X-ray techniques of the structures of important biochemical substances.” This recognized her work on penicillin and vitamin B12. She was only the third woman to win the Nobel Prize in Chemistry.
- Insulin Structure (1934-1969): After 35 years of work, Hodgkin and her team finally determined the structure of insulin, a hormone crucial for regulating blood sugar. This was a testament to her perseverance and dedication to solving complex scientific problems.
- Methodological Innovations:
- Improved Crystallographic Techniques: Hodgkin continuously refined and enhanced X-ray crystallographic techniques, pushing the possible limits.
- Patterson Function: She extensively used the Patterson function, a mathematical technique for analyzing X-ray diffraction data, which helped her interpret complex patterns.
- Computational Methods: She embraced the use of early computers to help with the massive calculations required for structure determination. This was crucial for solving the structures of large molecules like vitamin B12 and insulin.
- Heavy Atom Method: Developed and refined the “heavy atom method,” a technique for solving the phase problem in X-ray crystallography.
- Later Life and Legacy:
- Continued Research: She continued her research on the structures of biomolecules until late in her life.
- Mentorship: Mentored many students and researchers, training a generation of crystallographers.
- International Collaboration: Fostered international collaboration in science, working with scientists worldwide.
- Advocate for Peace and Social Justice: Was actively involved in peace movements and social justice issues.
- Numerous Awards and Honors: She received multiple awards and honors, including the Order of Merit (1965) and the Copley Medal of the Royal Society (1976).
- Death (1994): She died in 1994 at 84.
- Impact and Legacy:
- Pioneer of X-ray Crystallography: Transformed X-ray crystallography into a powerful tool for determining the structures of complex biomolecules.
- Revolutionized Structural Biology: Her structure determinations of penicillin, vitamin B12, and insulin had a profound impact on biology and medicine, enabling a deeper understanding of biological processes and the development of new drugs.
- Role Model for Women in Science: She was a role model for women in science, demonstrating that women could achieve groundbreaking success in a male-dominated field.
- Lasting Influence: Her techniques and approaches continue to be used and refined in structural biology today. Her work laid the foundation for modern structural biology and drug discovery.
- Inspiration: Inspired countless scientists.
Dorothy Crowfoot Hodgkin was a remarkable scientist whose perseverance, dedication, and technical brilliance revolutionized our understanding of the molecular world. Her work on the structures of penicillin, vitamin B12, and insulin were landmark achievements, and her pioneering contributions to X-ray crystallography had a profound and lasting impact on science and medicine. She was a true giant in the field, and her legacy inspires scientists today.
Dorothy Crowfoot Hodgkin, X-ray Crystallography, and Legacy. Table
Molecular structure of vitamin B12, as established by Hodgkin
(Wiki Image By NEUROtiker – Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=1672868)
Let’s detail the life, work, and legacy of Dorothy Crowfoot Hodgkin (1910-1994) in a table format, focusing on her pioneering use of X-ray crystallography to determine the structures of complex biomolecules.
| Aspect | Details | Significance |
| Early Life and Education | – Born in Cairo, Egypt (1910) to British parents. <br> – Early interest in science, particularly chemistry and crystals. <br> – Studied chemistry at Somerville College, Oxford, and later at the University of Cambridge (Newnham College) for her PhD. | – Overcoming significant gender barriers to pursuing a scientific career when few women were in science. <br> – Received a strong scientific foundation in chemistry and crystallography. |
| X-ray Crystallography Technique | – X-ray Diffraction: Learned and mastered the technique of X-ray crystallography, which involves shining X-rays through a crystal and analyzing the diffraction pattern produced. This pattern can be used to deduce the three-dimensional arrangement of atoms within the crystal. | – X-ray crystallography was a relatively new and challenging technique in the early 20th century. <br> – Hodgkin became a leading expert in this technique, pushing its limits to study increasingly complex molecules. |
| Key Structure Determinations | – Cholesterol Iodide (1940s): Early significant structure. <br> – Penicillin (1945): The structure of penicillin during World War II. This was a crucial breakthrough, enabling the mass production of this life-saving antibiotic. <br> – Vitamin B12 (1956): Determined the structure of vitamin B12, a very complex molecule essential for red blood cell formation and nerve function. This was a significant achievement in X-ray crystallography and earned her the Nobel Prize. <br> – Insulin (1969): After decades of work (starting in the 1930s), she finally determined the structure of insulin, a hormone crucial for regulating blood sugar. | – Revolutionized structural chemistry and biochemistry. <br> – Demonstrated the power of X-ray crystallography to determine the structures of complex biomolecules. <br> – Her work profoundly impacted medicine and biology. <br> – Penicillin: Enabled mass production during WWII, saving countless lives. <br> – Vitamin B12: Nobel Prize in Chemistry (1964); understanding its structure helped in treating pernicious anemia. <br> – Insulin: Crucial for understanding diabetes and improving treatments. |
| Methodological Innovations | – Improved Crystallographic Techniques: Developed and refined X-ray crystallographic techniques, pushing the possible limits. <br> – Patterson Function: Used and advanced the Patterson function, a mathematical technique for interpreting X-ray diffraction data. <br> – Computational Methods: Embraced using early computers to help with the complex calculations involved in structure determination. | – Made it possible to study increasingly complex molecules. <br> – Advanced the field of X-ray crystallography itself. |
| Mentorship and Collaboration | – Mentored many students and researchers: Trained and inspired a generation of crystallographers. <br> – Collaborated with scientists from around the world. | – Built a strong research group at Oxford. <br> – Fostered international collaboration in science. |
| Later Life and Recognition | – Continued her research until late in life. <br> – Numerous awards and honors, including the Nobel Prize in Chemistry (1964), the Order of Merit (1965), and the Copley Medal of the Royal Society (1976). <br> – Advocate for peace and social justice. | – Recognized as one of the most important scientists of the 20th century. <br> – A role model for women in science. |
| Legacy | – Pioneer of X-ray Crystallography: Transformed X-ray crystallography into a powerful tool for determining the structures of complex molecules. <br> – Impact on Biology and Medicine: Her structure determinations profoundly impacted biology and medicine, enabling a deeper understanding of biological processes and the development of new drugs. <br> – Inspiration to Scientists: Inspired generations of scientists, particularly women, to pursue careers in science. <br> – Continued Research: The techniques and approaches she pioneered continue to be used and refined in structural biology today. | – Her work laid the foundation for modern structural biology and drug discovery. <br> – She demonstrated the power of X-ray crystallography to reveal the secrets of life at the molecular level. <br> – A true scientific giant. |
Export to Sheets
Key Takeaways:
- Master of X-ray Crystallography: Hodgkin was a master of X-ray crystallography, pushing the technique to its limits to solve incredibly complex structures.
- Perseverance: Her work on insulin, in particular, demonstrated her extraordinary perseverance, taking over 30 years to complete.
- Impact on Medicine: Her structure determinations directly and significantly impacted medicine and human health.
- Role Model: She was a pioneering woman in science, achieving groundbreaking success in a male-dominated field.
This table comprehensively overviews Dorothy Crowfoot Hodgkin’s life, scientific contributions, and legacy. It highlights her pioneering work in X-ray crystallography, her determination of the structures of crucial biomolecules, and her impact on science and medicine. The format allows for a clear understanding of the progression of her career and the significance of her achievements.
Hodgkin X-ray Diffraction: Learned and mastered the technique of X-ray crystallography, which involves shining X-rays through a crystal and analyzing the diffraction pattern produced. Table.
Okay, let’s break down the technique of X-ray Crystallography and Dorothy Crowfoot Hodgkin’s mastery of it into a table format.
| Aspect of X-ray Crystallography | Description / Explanation |
| Technique Name | X-ray Crystallography (also known as X-ray Diffraction or XRD) |
| Purpose | To determine the precise three-dimensional arrangement of atoms within a crystalline solid, revealing the molecule’s structure, bond lengths, and bond angles. |
| Requirement: Crystallization | The substance being studied must first be grown into a well-ordered crystal. This means the molecules are arranged in a highly regular, repeating pattern (a crystal lattice). This step is often the most challenging, especially for large, complex biological molecules. |
| Basic Process: Diffraction | 1. A focused beam of X-rays is directed at the crystal. <br> 2. As the X-rays interact with the electrons of the atoms in the crystal lattice, they are scattered (diffracted) in specific directions. <br> 3. The scattered X-rays interfere with each other constructively and destructively, creating a unique diffraction pattern (a specific arrangement of spots of varying intensities). |
| Data Collection | The diffraction pattern (the positions and intensities of the diffracted spots) is recorded, historically using photographic film, now more commonly using electronic detectors. |
| Data Analysis: From Pattern to Structure | – The diffraction pattern contains encoded information about the arrangement of atoms in the crystal. <br> – Mathematical Analysis: Complex mathematical calculations (Fourier transforms) are used to convert the measured intensities of the diffraction spots into an electron density map. <br> – Phase Problem: A major hurdle is the “phase problem” – the diffraction experiment measures intensities but loses phase information, which is also needed. Hodgkin was skilled at using methods (like the heavy atom method or isomorphous replacement) to solve this. <br> – Model Building: The electron density map is interpreted to determine the positions of individual atoms, allowing scientists to build an accurate three-dimensional model of the molecule. |
| Hodgkin’s Role and Mastery | – Learned from Pioneers: Learned the technique from experts like J.D. Bernal. <br> – Master Practitioner: Became one of the world’s leading practitioners, known for her exceptional experimental skill, particularly in growing difficult crystals of complex biomolecules. <br> – Pushed Boundaries: Tackled molecules (penicillin, B12, insulin) that were significantly larger and more complex than what had previously been studied by X-ray crystallography. <br> – Meticulous Analysis: Was renowned for her careful and insightful analysis of complex diffraction data and electron density maps, often relying on painstaking calculations (initially manual, later aided by early computers). |
| Significance | – The definitive method for determining the precise atomic structure of crystalline materials. <br> – Revolutionized structural chemistry, biochemistry, and molecular biology. <br> – Provided the structural basis for understanding the function of countless molecules, including proteins, enzymes, DNA (by others), and drugs. <br> – Essential tool for rational drug design and materials science. |
Export to Sheets
In Summary:
Dorothy Hodgkin learned and masterfully applied the technique of X-ray crystallography, which involves analyzing the pattern produced when a crystal diffracts X-rays. Her exceptional skill in growing crystals and interpreting the complex diffraction data allowed her to determine the three-dimensional structures of vital and incredibly complex biological molecules like penicillin, vitamin B12, and insulin, profoundly impacting chemistry and medicine.
Hodgkin Cholesterol Iodide (1940s): Early significant structure. Table.
Okay, let’s create a table detailing Dorothy Hodgkin’s work on cholesteryl iodide, highlighting its significance as an early landmark structure determination using X-ray crystallography.
| Aspect of Cholesterol Iodide Work | Details / Explanation |
| Molecule Studied | Cholesteryl Iodide (An iodide derivative of cholesterol) |
| Time Period | Work started in the late 1930s, with significant progress and refinement into the early 1940s. |
| Technique Used | X-ray Crystallography |
| Goal | To determine the complete three-dimensional structure of a steroid molecule for the first time using X-ray diffraction. The complex, fused four-ring structure of steroids was known from chemical studies, but X-ray crystallography offered definitive proof of the spatial arrangement. |
| Challenges | – Molecular Complexity: While smaller than proteins like insulin, steroids represented a significant challenge for the crystallographic methods available at the time. <br> – Phase Problem: Solving the phase problem to translate diffraction intensities into an electron density map was a major hurdle. <br> – Computational Intensity: Required laborious manual calculations before the advent of powerful computers. |
| Key Achievement | Successfully determined the complete 3D structure of cholesteryl iodide. This was achieved largely through the analysis of Patterson maps, which could be calculated directly from the intensities and were particularly useful because of the presence of the heavy iodine atom. |
| Significance | – Early Landmark Structure: One of the first complex organic molecules of biological significance whose three-dimensional structure was fully determined by X-ray crystallography. <br> – Confirmed Steroid Structure: Provided definitive, unambiguous proof of the fused-ring structure common to cholesterol and other steroid molecules. <br> – Validated X-ray Crystallography: Demonstrated the power and potential of X-ray crystallography for solving complex organic structures, building confidence in the technique. <br> – Methodological Development: Contributed to the development and refinement of crystallographic methods, particularly the use of the Patterson function and heavy-atom methods. <br> – Stepping Stone: This success, alongside her concurrent work on penicillin, was an important stepping stone towards tackling even larger biological macromolecules like Vitamin B12 and insulin. |
Export to Sheets
In Summary:
Dorothy Hodgkin’s determination of the structure of cholesteryl iodide in the early 1940s was a significant early success in her career and for the field of X-ray crystallography. It proved the technique’s capability for revealing the detailed architecture of complex organic molecules, confirmed the fundamental structure of steroids, and paved the way for her later, even more challenging, structure determinations of vital biomolecules.
Hodgkin Penicillin (1945): The structure of penicillin during World War II. This was a crucial breakthrough, enabling the mass production of this life-saving antibiotic. Table.
Okay, let’s create a table detailing Dorothy Hodgkin’s crucial work on determining the structure of penicillin during World War II.
| Aspect of Penicillin Structure Work | Details / Explanation |
| Molecule Studied | Penicillin (specifically, various penicillin salts like potassium and sodium penicillin) |
| Time Period | Primarily 1942-1945 (During World War II) |
| Context | – Penicillin was discovered by Fleming and developed as an antibiotic by Florey & Chain. <br> – Highly effective against bacterial infections, making it vital for the war effort (treating wounded soldiers). <br> – Urgent Need: Demand far outstripped supply; understanding the structure was critical for improving production or enabling chemical synthesis. <br> – Structural Debate: Chemists debated the correct structure, particularly the presence of an unusual, highly strained four-membered beta-lactam ring. |
| Technique Used | X-ray Crystallography |
| Research Effort | This was a major, highly secret, transatlantic collaborative effort involving research groups in the UK (led by Hodgkin at Oxford) and the US. Hodgkin’s group played the central role in the structural determination using X-ray methods. |
| Challenges | – Crystallization: Obtaining stable, suitable crystals of penicillin salts was difficult. <br> – Molecular Complexity: Penicillin was a complex organic molecule for the crystallographic techniques and computational resources available at the time. <br> – Wartime Urgency & Secrecy: Research conducted under pressure and secrecy due to its military importance. |
| Key Achievement | Dorothy Hodgkin and her team successfully determined the complete three-dimensional arrangement of atoms in penicillin, definitively confirming the presence of the controversial beta-lactam ring. |
| Methodology | – Utilized X-ray diffraction patterns from crystals of various penicillin salts. <br> – Required extensive and laborious calculations (largely manual or with early computational aids) to analyze the diffraction data and produce electron density maps. <br> – Interpreted the electron density maps to build the final 3D model. |
| Significance | – Crucial Breakthrough: Settled the intense scientific debate about penicillin’s correct structure. <br> – Enabled Improved Production: While full chemical synthesis proved too difficult for immediate mass production, knowing the correct structure was essential for understanding its chemical properties, degradation pathways, and for optimizing the fermentation processes used to produce it naturally from mold, leading to increased yields. <br> – Foundation for Synthetic Variants: Understanding the core structure (especially the beta-lactam ring) was fundamental for the later development of semi-synthetic penicillins with improved effectiveness and broader applications. <br> – Impact on WWII: Contributed significantly to the Allied war effort by helping make this life-saving antibiotic more widely available. <br> – Advanced Crystallography: Further cemented X-ray crystallography as a powerful tool for determining the structures of complex biological molecules. |
Export to Sheets
In Summary:
Dorothy Hodgkin’s determination of the penicillin structure using X-ray crystallography during World War II was a critical scientific achievement. It resolved a major chemical debate and, crucially, provided the structural knowledge necessary to significantly improve the production of this life-saving antibiotic, directly impacting the war effort and revolutionizing medicine. This success further highlighted the power of her chosen technique.
Hodgkin Vitamin B12 (1956): The structure of vitamin B12 is a very complex molecule essential for red blood cell formation and nerve function. This was a significant achievement in X-ray crystallography and earned her the Nobel Prize. Table.
Okay, let’s create a table detailing Dorothy Hodgkin’s groundbreaking work on determining the structure of Vitamin B12 (Cobalamin).
| Aspect of Vitamin B12 Structure Work | Details / Explanation |
| Molecule Studied | Vitamin B12 (Cobalamin) |
| Time Period | Research spanned from the late 1940s, with the structure determined and published around 1955-1956. |
| Biological/Medical Context | – Vitamin B12 is essential for human health, particularly for red blood cell formation, neurological function, and DNA synthesis. <br> – Deficiency leads to pernicious anemia, a severe and previously fatal condition. <br> – Understanding its structure was crucial for understanding its function and deficiency diseases. |
| Technique Used | X-ray Crystallography |
| Challenges | – Extreme Molecular Complexity: Vitamin B12 was significantly larger and more structurally complex than any non-polymeric molecule whose structure had been determined before (~100 non-hydrogen atoms). It contains a central cobalt atom within a complex, large corrin ring system. <br> – Computational Intensity: The analysis required processing vast amounts of diffraction data and performing extremely complex calculations, pushing the limits of early electronic computers (like EDSAC in Cambridge and SWAC in California, used in collaboration). <br> – Phase Problem: Solving the phase problem for such a large, complex, non-centrosymmetric molecule was a major crystallographic hurdle. |
| Key Achievement | Dorothy Hodgkin and her international team successfully determined the complete three-dimensional structure of vitamin B12, revealing its intricate architecture. |
| Methodology | – Utilized X-ray diffraction data from crystals of vitamin B12. <br> – Leveraged the presence of the relatively heavy cobalt atom to help solve the phase problem using heavy-atom methods and Patterson analysis. <br> – Required international collaboration and the use of the most advanced computational resources available at the time. <br> – Involved years of meticulous data collection and interpretation. |
| Significance | – Landmark Achievement in Crystallography: Solving the B12 structure was a monumental achievement, proving that X-ray crystallography could determine the structures of even very large and complex biomolecules. <br> – Understanding Vitamin B12 Function: Revealed the intricate structure, providing the basis for understanding its unique biochemical mechanisms and its role in biological processes. <br> – Nobel Prize in Chemistry (1964): This specific achievement was the primary reason Dorothy Hodgkin was awarded the Nobel Prize in Chemistry, recognizing the immense difficulty and importance of the work. <br> – Advanced Computational Chemistry: The complexity of the B12 analysis spurred advancements in computational methods used in crystallography. |
Export to Sheets
In Summary:
Dorothy Hodgkin’s determination of the Vitamin B12 structure was a pinnacle of achievement in X-ray crystallography. It involved tackling a molecule of unprecedented complexity for its time, requiring immense skill, perseverance, international collaboration, and early computers. This groundbreaking work revealed the structure of a vital biological molecule, essential for understanding pernicious anemia, and cemented the power of X-ray crystallography, which was the central accomplishment recognized by her 1964 Nobel Prize in Chemistry.
Hodgkin Insulin (1969): After decades of work (starting in the 1930s), she finally determined the structure of insulin, a hormone crucial for regulating blood sugar. Table.
Okay, let’s create a table detailing Dorothy Hodgkin’s decades-long work culminating in the determination of the structure of insulin.
| Aspect of Insulin Structure Work | Details / Explanation |
| Molecule Studied | Insulin |
| Biological/Medical Importance | A vital hormone produced by the pancreas that regulates blood sugar levels by facilitating the uptake of glucose from the blood into cells. Its deficiency or improper function causes diabetes mellitus. Understanding its structure was critical for understanding its mechanism and treating diabetes. |
| Time Period | An exceptionally long and persistent research effort spanning 35 years: <br> – Began research: 1934 (obtained first X-ray diffraction photographs of insulin crystals shortly after graduating). <br> – Structure finally determined and published: 1969. |
| Context & Early Challenges | – Insulin was one of the first proteins subjected to X-ray crystallographic study. <br> – Proteins are large, complex macromolecules, making structural determination vastly more difficult than for smaller molecules like penicillin or even vitamin B12. <br> – Techniques for protein crystallography were in their infancy in the 1930s. <br> – Growing suitable crystals of insulin was extremely difficult and took years to perfect. |
| Technique Used | X-ray Crystallography |
| Major Hurdles Overcome | – Protein Crystallization: Years spent optimizing methods to grow insulin crystals large enough and ordered enough for high-resolution diffraction. <br> – Phase Problem: Solving the phase problem for a molecule the size of insulin was a major theoretical and computational challenge. Required advancements in heavy-atom methods (isomorphous replacement). <br> – Computational Power: Required significant advancements in computational methods and the use of early electronic computers to process the vast amount of diffraction data and perform the complex calculations needed for structure determination. |
| Key Achievement | After 35 years of dedicated effort, Dorothy Hodgkin and her team successfully determined the complete three-dimensional atomic structure of insulin. |
| Methodology | – Utilized X-ray diffraction data collected from insulin crystals (often zinc insulin). <br> – Required incremental progress over decades as crystallographic techniques, phasing methods (isomorphous replacement), and computational power advanced. <br> – Involved meticulous data analysis and interpretation of complex electron density maps. |
| Significance | – Culmination of a Lifelong Quest: Represented the fulfillment of a scientific goal Hodgkin had pursued since the very beginning of her independent research career. <br> – Testament to Perseverance: An extraordinary example of scientific dedication and perseverance over decades. <br> – Understanding Insulin Function: Provided the detailed structural basis for understanding how insulin binds to its receptor and performs its vital role in glucose regulation. <br> – Impact on Diabetes Research: Foundational for modern diabetes research, enabling the rational design of insulin analogs and improved treatments. <br> – Landmark in Structural Biology: A major milestone in the history of protein crystallography and structural biology, demonstrating that the structures of important protein hormones could be determined. |
Export to Sheets
In Summary:
Dorothy Hodgkin’s determination of the insulin structure in 1969, after 35 years of relentless work, stands as one of the great achievements in structural biology. It required overcoming immense technical challenges related to protein crystallization, X-ray diffraction analysis, and computation. This landmark success not only revealed the architecture of a hormone crucial for treating diabetes but also showcased Hodgkin’s extraordinary scientific skill and unwavering perseverance.
Hodgkin Improved Crystallographic Techniques: Developed and refined X-ray crystallographic techniques, pushing the possible limits. Table.
Okay, let’s create a table focusing on how Dorothy Crowfoot Hodgkin developed, refined, and pushed the limits of X-ray Crystallographic techniques during her career. While she didn’t invent the core principles, her application of the method to increasingly complex molecules necessitated significant advancements and showcased its potential.
| Aspect of Technique Improvement | Details / How Hodgkin Contributed | Significance |
| 1. Tackling Unprecedented Complexity | X-ray crystallography was applied to molecules far larger and more complex than previously thought possible, starting with cholesterol iodide, moving to penicillin, then the vastly more complex Vitamin B12, and culminating in the protein insulin. | Demonstrated the power and potential of X-ray crystallography for biology and medicine. Her successes silenced skeptics and proved the technique could unravel the structures of life’s essential molecules, driving the field forward. |
| 2. Mastery & Refinement of Phasing Methods | Became a leading expert in using and refining methods to solve the “phase problem” (determining the phases of diffracted X-rays, which are lost in experiments but needed for structure calculation). She made masterful use of: <br> – Patterson Function Analysis: Interpreting maps derived directly from intensities, crucial for locating heavy atoms. <br> – Heavy Atom Method / Isomorphous Replacement: Skillfully incorporating heavy atoms (like iodine, cobalt, or other metals) into crystals to help determine the phases. | Made solving complex, non-centrosymmetric structures feasible. Her expertise in phasing was critical for determining the structures of Vitamin B12 and insulin. |
| 3. Early Adoption of Computational Methods | Recognized early on that the calculations required for complex structures were beyond manual capacity. She collaborated with mathematicians and utilized some of the earliest electronic computers (e.g., EDSAC, SWAC) to process diffraction data and perform Fourier calculations for Vitamin B12 and insulin. | Pioneered the use of computers in structural biology. Her work highlighted the essential role that computation would play in solving large molecular structures and helped drive advancements in computational crystallography. |
| 4. Advancements in Crystallization | Known for her exceptional patience and skill in growing high-quality single crystals of difficult biological molecules (especially insulin), often requiring years of experimentation with different conditions. | Crucial Experimental Skill: Obtaining suitable crystals is often the bottleneck in crystallography. Hodgkin’s mastery of this experimental art was fundamental to her ability to collect usable diffraction data for complex molecules. |
| 5. Meticulous Data Collection & Analysis | Renowned for her extreme care and thoroughness in collecting X-ray diffraction data and her painstaking, detailed interpretation of complex diffraction patterns and electron density maps. | Ensured the accuracy and reliability of her groundbreaking structure determinations. Set a high standard for experimental rigor in a field where precise data is paramount. |
| 6. Pushing Resolution Limits | Consistently worked towards achieving higher resolution diffraction data for the molecules she studied. | Provided more detailed structural information. Higher resolution allows for more precise determination of atomic positions, bond lengths, angles, and subtle structural features. |
Export to Sheets
In Summary:
Dorothy Hodgkin was not just a user of X-ray crystallography; she was a crucial figure in developing its potential. By tackling molecules of unprecedented complexity, she forced the refinement of existing methods (like phasing techniques) and spurred the adoption of new tools (like computers). Her experimental skill, particularly in crystallization, combined with her meticulous analytical approach, allowed her to push the boundaries of what was possible with X-ray diffraction, transforming it into the powerful tool for structural biology that it is today. She demonstrated the technique’s power and inspired others to apply it to even more challenging biological systems.
Hodgkin Patterson Function: Used and advanced the Patterson function, a mathematical technique for interpreting X-ray diffraction data. Table.
Okay, let’s create a table focusing on Dorothy Hodgkin’s use and advancement of the Patterson function within the context of X-ray crystallography.
| Aspect of Patterson Function Use by Hodgkin | Details / Explanation |
| Technique Name | Patterson Function (also Patterson Synthesis or Patterson Map) |
| Developer | Arthur Lindo Patterson (developed in 1934) |
| Mathematical Basis | A mathematical technique (a type of Fourier transform) is applied directly to the intensities of the measured X-ray diffraction spots. Unlike the electron density map, it does not require phase information (which is lost in the experiment). |
| What it Represents | The Patterson map displays vectors between pairs of atoms within the crystal’s unit cell. Peaks in the Patterson map correspond to these interatomic vectors, and the height of a peak is related to the product of the electron numbers of the two atoms connected by that vector. |
| Solving the “Phase Problem” | The primary challenge in X-ray crystallography is the “phase problem” (loss of phase information). The Patterson function helps overcome this, especially when used with the heavy atom method. |
| Hodgkin’s Application (Heavy Atom Method) | – Hodgkin became a master practitioner of using Patterson maps, particularly when her crystals contained one or more heavy atoms (atoms with significantly more electrons than carbon, nitrogen, oxygen, e.g., iodine, cobalt, or other metals). <br> – Locating Heavy Atoms: Because heavy atoms have many electrons, the vectors between these heavy atoms produce very strong, distinct peaks in the Patterson map. Hodgkin excelled at the difficult task of interpreting these complex maps to pinpoint the positions of the heavy atoms within the unit cell. |
| Bootstrapping Structure Solution | – Once the positions of the heavy atoms were determined from the Patterson map, their contribution to the phases of the diffraction spots could be calculated. <br> – This initial phase information (from the heavy atoms) served as a crucial starting point to iteratively determine the phases of all the reflections, ultimately allowing the calculation of a complete electron density map and the determination of the full molecular structure. |
| Examples in Hodgkin’s Work | – Cholesteryl Iodide: The position of the heavy iodine atom was key, and it was found using Patterson methods. <br> – Penicillin: Patterson’s analysis of data from various heavy-atom salts (potassium, rubidium) was essential. <br> – Vitamin B12: The central cobalt atom was the crucial heavy atom; interpreting the B12 Patterson map was a major analytical feat that enabled the structure solution. <br> – Insulin: While requiring more advanced isomorphous replacement techniques (using multiple heavy atom derivatives), Patterson methods were still vital for locating the heavy atoms in each derivative. |
| Significance of Hodgkin’s Contribution | – Enabled Complex Structure Solution: Her skillful application and interpretation of Patterson maps were essential for solving the phase problem for the complex, non-centrosymmetric structures she tackled (penicillin, B12, insulin). <br> – Pushed Methodological Limits: She demonstrated the power of the Patterson function beyond simpler structures, showing its applicability to large biological molecules and advancing the practical use of the heavy atom method. <br> – Advanced the Field: Her success using these techniques contributed significantly to developing and refining crystallographic methodology. |
Export to Sheets
In Summary:
Dorothy Hodgkin did not invent the Patterson function, but she was one of its most skilled and influential practitioners. She masterfully used it, particularly in conjunction with heavy atoms, to overcome the critical phase problem in X-ray crystallography. Her ability to interpret complex Patterson maps was fundamental to her success in determining the groundbreaking structures of penicillin and Vitamin B12, thereby significantly advancing the technique and demonstrating its power for solving complex biological structures.
Hodgkin Computational Methods: Embraced using early computers to help with the complex calculations involved in structure determination. Table.
Okay, let’s create a table focusing on Dorothy Hodgkin’s pioneering embrace and use of early computational methods in X-ray crystallography.
| Aspect of Computational Method Use | Details / Explanation |
| Context: The Computational Bottleneck | – Determining molecular structures from X-ray diffraction data involves immense and complex calculations, primarily Fourier transforms, to generate electron density maps from phased diffraction intensities. <br> – For simpler structures, these could be done manually using slide rules, mechanical calculators (e.g., Marchant calculators), and specialized analogue aids (e.g., Beevers-Lipson strips). <br> – For complex molecules like Vitamin B12 and proteins, manual calculation was prohibitively slow, laborious, and prone to error, representing a major bottleneck in structure determination. |
| Emergence of Early Computers | – The late 1940s and 1950s saw the development of the first electronic digital computers (e.g., EDSAC in Cambridge, SWAC in California, Manchester Mark 1). These machines offered the potential to automate and drastically speed up complex calculations. |
| Hodgkin’s Recognition and Early Adoption | – As she tackled the extremely complex structure of Vitamin B12, Hodgkin recognized that the sheer volume of calculation required was exceeding the limits of manual methods. <br> – She was one of the first crystallographers to actively embrace the potential of these new electronic computers to overcome the computational bottleneck. |
| Collaboration with Computational Experts | – Hodgkin was not a computer programmer herself, but she actively sought out collaborations with mathematicians and early computer scientists who could develop and implement the necessary crystallographic programs. <br> – Vitamin B12: Crucial calculations were performed using SWAC (Standards Western Automatic Computer) in California (in collaboration with Kenneth Trueblood) and EDSAC (Electronic Delay Storage Automatic Calculator) in Cambridge. <br> – Insulin: Later work on insulin relied even more heavily on advancing computational power and algorithms. |
| Impact of Computers on Her Work | – Enabled Solution of Complex Structures: Computers made it feasible to perform the vast number of calculations required to solve the structures of Vitamin B12 and insulin within a reasonable timeframe. These solutions might have been practically impossible otherwise. <br> – Increased Speed and Accuracy: Dramatically sped up the calculation process and reduced the potential for human error compared to manual methods. <br> – Facilitated Refinement: Allowed for more complex refinement calculations to improve the accuracy of the final atomic models. |
| Hodgkin’s Role in Computational Application | – Visionary Adoption: Recognized the transformative potential of computing for crystallography early on. <br> – Driving Force: Her challenging structural problems (like B12) provided a strong motivation for developing computational crystallographic methods. <br> – Data Provider and Interpreter: Provided the high-quality experimental data for the computations and expertly interpreted the resulting electron density maps. |
| Significance | – Pioneered Computational Crystallography: Hodgkin’s work demonstrated computers’ essential role in modern structural biology and chemistry. <br> – Bridged Experiment and Computation: Showcased the power of combining meticulous experimental work with advanced computational analysis. <br> – Made Landmark Discoveries Possible: Computational methods were indispensable tools that enabled her landmark structure determinations of Vitamin B12 and insulin. |
Export to Sheets
In Summary:
Dorothy Hodgkin was a visionary scientist who recognized the limitations of manual calculation for solving complex molecular structures. She became an early and crucial adopter of electronic computers in X-ray crystallography, collaborating with computational experts to perform the immense calculations required for her landmark Vitamin B12 and insulin determinations. Her embrace of computational methods enabled her groundbreaking discoveries. It helped pioneer the field of computational crystallography, demonstrating the indispensable role computers would play in unraveling the structures of life’s molecules.
Robert Burns Woodward; Organic Synthesis

Robert Burns Woodward
(Wiki Image By Louis Fabian Bachrach Jr. (photographer), via The Nobel Foundation – http://www.nobelprize.org/nobel_prizes/chemistry/laureates/1965/, PD-Sweden, https://en.wikipedia.org/w/index.php?curid=34664939)
Robert Burns Woodward Quotes. Table
Let’s organize a selection of quotes from Robert Burns Woodward, or representative statements reflecting his known views, into a table format. Woodward was renowned for his precision in chemistry. While perhaps less known for widespread aphorisms than someone like Pauling, his statements often reflect his rigorous approach to organic synthesis and chemical structure.
| Quote Number | Quote | Source | Significance/Explanation |
| 1 | “Organic synthesis is a science, but it is also an art.” | Widely attributed; reflects his core philosophy | Captures Woodward’s view that creating complex molecules requires scientific knowledge, logic, creativity, intuition, and strategic elegance. |
| 2 | “Structure is everything in organic chemistry.” | Attributed / Representative | Highlights the fundamental importance he placed on understanding the precise three-dimensional arrangement of atoms in a molecule, which dictates its properties and reactivity. |
| 3 | “The key to successful synthesis lies in rigorous analysis and strategic planning.” | The representative summarizes his approach. | He emphasizes his meticulous methodology—carefully planning each step of a complex synthesis, anticipating potential problems, and devising innovative solutions. |
| 4 | “It has seemed interesting to me that the human mind, looking only at the structure of the molecule, could create such a complex sequence of reactions.” | Paraphrased from his Nobel Lecture (1965) | Reflects his awe and intellectual satisfaction regarding the power of organic chemistry to construct intricate natural products through logical, multi-step reaction sequences designed purely from understanding structure. |
| 5 | “[These simple orbital symmetry rules]… provided a basis for understanding… a wide variety of concerted reactions.” | Representative; summarizing the impact of the Woodward-Hoffmann rules | Highlights the significance of the Woodward-Hoffmann rules (developed with Roald Hoffmann) in providing a robust theoretical framework for predicting the outcomes of an important class of organic reactions (pericyclic reactions). |
| 6 | “The synthesis of complex molecules provides a unique intellectual challenge.” | Representative | He reflects on his view of total synthesis as a demanding intellectual puzzle that requires problem-solving skills, deep knowledge, and creativity. |
Export to Sheets
Key Takeaways from the Quotes:
- Synthesis as Art and Science: Woodward saw organic synthesis as requiring rigorous scientific knowledge and creative artistry.
- Primacy of Structure: He emphasized the fundamental importance of molecular structure in determining chemical behavior.
- Strategic Planning: His work was characterized by meticulous planning and elegant strategic design.
- Intellectual Challenge: He viewed the construction of complex molecules as a profoundly satisfying intellectual endeavor.
- Theory and Practice: He contributed significantly to the synthesis practice and the underlying theoretical principles (orbital symmetry rules).
This table provides a structured overview of key statements reflecting Robert Burns Woodward’s philosophy and contributions. It showcases his focus on the elegance and intellectual rigor of organic synthesis, his emphasis on structure, and his role in developing theoretical tools to understand chemical reactions. The quotes reveal a mind fascinated by the challenge and beauty of building complex molecules.
Robert Burns Woodward YouTube Video
- R. B. Woodward, chemistry legend by Chemical & Engineering News: 21,352 views (http://www.youtube.com/watch?v=YewUnM0hXs4)
- Robert Burns Woodward by Cm: 1,358 views (http://www.youtube.com/watch?v=s4Ahuinwe7A)
- Part 1 B12 Lecture RBW Woodward 11-27-72 by Thomas Hoye: 461 views (http://www.youtube.com/watch?v=CSNEs01dwsY)
- Robert Burns Woodward | Wikipedia audio article by Wikipedia tts: 277 views (http://www.youtube.com/watch?v=2_dZPyYTdQk)
- Robert Burns Woodward (EN) by about-the-world: 20 views (http://www.youtube.com/watch?v=9-iaVx4PaAU)
Robert Burns Woodward History
Woodward talked about Chlorophyll in 1965
(Wiki Image By Peter Geymayer at German Wikipedia / (Original text: Dr. Peter Geymayer) – Self-photographed, Public Domain, https://commons.wikimedia.org/w/index.php?curid=3751318)
Robert Burns Woodward (1917-1979) was an American organic chemist widely considered the preeminent organic chemist of the 20th century. He is best known for his spectacular achievements in the total synthesis of complex organic molecules, his contributions to developing reaction mechanisms, and his work (with Roald Hoffmann) on the orbital symmetry rules governing pericyclic reactions (the Woodward-Hoffmann rules).
- Early Life and Education (1917-1937):
- Born in Boston, Massachusetts (1917), He showed an extraordinary aptitude for chemistry from a very young age. He reportedly completed the experiments in an elementary school’s standard organic chemistry textbook.
- Massachusetts Institute of Technology (MIT): He entered MIT at 16 and progressed through the undergraduate curriculum astonishingly.
- Ph.D. at MIT (1937): He earned his Ph.D. in chemistry from MIT at the age of 20. His doctoral work was remarkably independent, reflecting his early brilliance.
- Early Career and Total Synthesis (1937-1960s):
- Harvard University: He spent almost his entire career at Harvard University, becoming a professor at a young age.
- World War II: During World War II, he consulted on various war-related projects, including synthesizing quinine (an antimalarial drug).
- Total Synthesis: Woodward’s primary focus was on total synthesis, which is the complete chemical synthesis of complex organic molecules, often natural products, from simple, commercially available starting materials. This was considered an incredibly challenging and intellectually demanding area of chemistry.
- Strategic Planning: His syntheses were characterized by meticulous planning, elegant strategies, and the innovative use of new reactions and reagents. He approached synthesis like a complex, multi-step chess game, carefully considering each step and its consequences.
- “Art and Science” of Synthesis: He viewed organic synthesis as both an art and a science requiring creativity and rigorous logic.
- Key Total Syntheses (Early Career):
- Quinine (1944): While the formal synthesis had been claimed earlier, Woodward completed the full synthesis. This was a significant achievement due to quinine’s importance as an antimalarial drug.
- Cholesterol and Cortisone (1951): Synthesized these complex steroids, demonstrating the power of organic synthesis to create molecules with important biological activity.
- Reserpine (1956): Synthesized reserpine, a complex alkaloid with important medicinal properties (used as a tranquilizer).
- Chlorophyll (1960): Synthesized chlorophyll, the green pigment responsible for photosynthesis. This was a major achievement, demonstrating his mastery of complex molecule synthesis.
- Woodward-Hoffmann Rules (1965):
- Collaboration with Roald Hoffmann: Collaborated with theoretical chemist Roald Hoffmann to develop the Woodward-Hoffmann rules (also known as the rules of orbital symmetry).
- Pericyclic Reactions: These rules predict the stereochemistry (three-dimensional arrangement of atoms) of pericyclic reactions, a class of organic reactions that involve a cyclic transition state.
- Orbital Symmetry: The rules are based on the symmetry properties of molecular orbitals and how they interact during a reaction.
- Significance: The Woodward-Hoffmann rules provided a robust theoretical framework for understanding and predicting the outcome of a large class of organic reactions. They unified theory and experiment, providing a deep understanding of reaction mechanisms.
- Vitamin B12 Synthesis and Later Career (1970s):
- Vitamin B12 Synthesis (1972): Completed the total synthesis of vitamin B12, a monumental achievement in organic synthesis. This was a collaborative effort with Albert Eschenmoser’s group in Switzerland, involving over 100 researchers spanning over a decade. The synthesis was incredibly complex, involving nearly 100 individual steps.
- Continued Research: Continued to work on challenging synthetic problems and to develop new synthetic methods.
- Awards and Recognition:
- Nobel Prize in Chemistry (1965): Awarded the Nobel Prize in Chemistry “for his outstanding achievements in the art of organic synthesis.” The Nobel committee specifically cited his syntheses of complex natural products.
- Numerous other awards: He received numerous other prestigious awards and honors throughout his career.
- Death and Legacy:
- Death (1979): He died in 1979 at 62.
- “The Preeminent Organic Chemist of the 20th Century”: Widely regarded as the greatest organic chemist of his time, and perhaps of all time.
- Transformed Organic Synthesis: His work transformed organic synthesis from a largely empirical field into a more rational and intellectually rigorous discipline.
- Strategic Planning: He emphasized the importance of strategic planning in organic synthesis, developing elegant and innovative approaches to complex molecular targets.
- Inspiration to Chemists: His syntheses are considered works of art, admired for their beauty, ingenuity, and intellectual depth. They continue to inspire generations of organic chemists.
- Impact on Drug Discovery: His work on the synthesis of complex molecules paved the way for the development of new drugs and materials.
- Woodward-Hoffmann Rules: These rules remain fundamental in organic chemistry, providing a powerful tool for understanding and predicting reaction outcomes.
Robert Burns Woodward was a towering figure in 20th-century chemistry. His achievements in total synthesis were unparalleled, and his contributions to theoretical organic chemistry were profound. He combined exceptional synthetic skill with deep theoretical understanding, transforming the field of organic chemistry and leaving a lasting legacy of intellectual brilliance and chemical artistry. He was a master of molecular architecture, creating complex structures with precision and elegance.
Robert Burns Woodward, Organic Synthesis, and Legacy. Table
Reflux apparatus
(Wiki Image By Tooto (talk) (Uploads) – Own work, Public Domain, https://commons.wikimedia.org/w/index.php?curid=109791648)
Let’s present the life, work, and legacy of Robert Burns Woodward (1917-1979), focusing on his contributions to organic synthesis in a structured table format. This will highlight his key achievements and their significance.
| Aspect | Details | Significance |
| Early Life and Education | – Born in Boston, Massachusetts (1917). <br> – Prodigy in chemistry; entered MIT at age 16 and earned a PhD at age 20. | – Demonstrated exceptional talent and early promise in chemistry. |
| Academic Career | – He spent most of his career at Harvard University, becoming a professor at a young age. | – Established a leading center for organic chemistry research at Harvard. |
| Key Contributions: Total Synthesis | – Master of Total Synthesis: Considered the preeminent organic chemist of the 20th century, known for his mastery of total synthesis – the complete chemical synthesis of complex organic molecules from simple, commercially available starting materials. <br> – Strategic Planning: His syntheses were characterized by elegant and innovative strategies, involving careful planning, the use of novel reactions, and a deep understanding of reaction mechanisms. He approached synthesis like a complex chess game. <br> – Key Syntheses: Achieved the total synthesis of many complex natural products, including: <br> – Quinine (1944) – an anti-malarial drug <br> – Cholesterol and Cortisone (1951) – steroids <br> – Reserpine (1956) – a tranquilizer <br> – Chlorophyll (1960) – the pigment responsible for photosynthesis <br> – Vitamin B12 (1972) – a very complex molecule, a collaborative effort with Albert Eschenmoser | – Revolutionized organic synthesis. Demonstrated that even incredibly complex organic molecules could be synthesized in the laboratory. <br> – Pushed the boundaries of what was considered possible in organic synthesis. His syntheses involved making molecules and developing new organic synthesis methods and strategies. <br> – Inspired generations of organic chemists. |
| Key Contributions: Theoretical Organic Chemistry | – Woodward-Hoffmann Rules (1965): Developed (with Roald Hoffmann) the Woodward-Hoffmann rules, which predict the stereochemistry (three-dimensional arrangement of atoms) of pericyclic reactions (a class of organic reactions). These rules are based on the symmetry properties of molecular orbitals. | – Provided a robust theoretical framework for understanding and predicting the outcome of a large class of organic reactions. <br> – Unified theory and experiment. The rules were based on theoretical principles but had immediate practical applications for synthetic chemists. |
| Key Contributions: Spectroscopy | Significant contributions were made to using physical methods, particularly ultraviolet-visible spectroscopy, to determine the chemical structure. | Helped to establish a powerful tool. |
| Awards and Recognition | – Nobel Prize in Chemistry (1965): Awarded “for his outstanding achievements in the art of organic synthesis.” <br> – Numerous other awards and honors. | – Recognized as one of the most outstanding organic chemists of all time. |
| Teaching and Mentorship | – Trained and mentored many students who became leading organic chemists. | – He significantly impacted the development of organic chemistry by influencing future generations of scientists. |
| Legacy | – Transformed Organic Synthesis: His work transformed organic synthesis from a largely empirical art into a more rational and predictable science. <br> – Inspiration to Chemists: His syntheses are considered works of art, inspiring generations of chemists with their elegance and ingenuity. <br> – Impact on Drug Discovery: His work on synthesizing complex molecules paved the way for developing new drugs and materials. <br> – Woodward-Hoffmann Rules: These rules remain fundamental in organic chemistry. <br> – Emphasis on Strategic Planning: His emphasis on strategic planning and developing new synthetic methods continues to influence the field. | – His work continues to be studied and admired by organic chemists today. <br> – He is considered a giant in the field, and his legacy is secure. |
Export to Sheets
Key Takeaways:
- Master of Synthesis: Woodward was a master of organic synthesis, achieving the seemingly impossible synthesis of incredibly complex molecules.
- Strategic Thinker: He was a brilliant strategic thinker, planning his syntheses meticulously and developing new methods to overcome challenges.
- Theoretical Contributions: He also made significant theoretical contributions, particularly the Woodward-Hoffmann rules.
- Lasting Impact: His work transformed organic synthesis and continues to influence today’s field.
This table provides a comprehensive overview of Robert Burns Woodward’s life, his scientific contributions (particularly in organic synthesis), and his lasting legacy. It highlights his key achievements, innovative approach to synthesis, and impact on organic chemistry. The format allows for a clear understanding of the progression of his career and the significance of his work.
Woodward, Master of Total Synthesis: Woodward is considered the preeminent organic chemist of the 20th century. He is known for mastering total synthesis, the complete chemical synthesis of complex organic molecules from simple, commercially available starting materials. Table.
Vitamin B12 total synthesis: Retrosynthetic analysis of the Woodward–Eschenmoser total synthesis reported in two variants by these groups in 1972. The work involved more than 100 PhD trainees and postdoctoral fellows from 19 countries. The retrosynthesis presents the disassembly of the target vitamin in a manner that makes chemical sense for its eventual forward construction. The target, Vitamin B12 (I), is envisioned to be prepared by simply adding its tail, which had earlier been shown to be feasible. The needed precursor, cobyric acid (II), then becomes the target and constitutes the “corrin core” of the vitamin. Its preparation was envisaged to be possible via two pieces, a “western” part composed of the A and D rings (III) and an “eastern” part composed of the B and C rings (IV). The restrosynthetic analysis then envisions the starting materials required to make these two complex parts, the yet complex molecules V–VIII.
(Wiki Image By Dissolution – Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=10669897)
Okay, let’s create a table outlining the key elements that define Robert Burns Woodward as the “Master of Total Synthesis.”
| Aspect of Mastery | Details / Explanation |
| Reputation | Widely considered the preeminent organic chemist of the 20th century. |
| Area of Expertise | Total Synthesis: His primary focus and area of exceptional skill. |
| Definition of Total Synthesis | The complete chemical synthesis of complex organic molecules (often natural products) starts from simple, readily available (commercially available) precursor materials through a sequence of chemical reactions. |
| Key Characteristics of Work | Molecular Complexity: Successfully synthesized molecules of extraordinary structural complexity, such as steroids (cholesterol, cortisone), alkaloids (reserpine), pigments (chlorophyll), and vitamins (Vitamin B12). |
| Approach & Methodology | Characterized by elegant and innovative strategies involving careful planning, the creative use of known and novel reactions, and a deep understanding of reaction mechanisms. He approached synthesis like a complex chess game. |
| Impact | Demonstrated the Power of Synthesis: Proved that chemists could recreate nature’s most complex molecules. <br> Advanced Synthetic Methodology: His work often led to the development or refinement of new synthetic methods. <br> Inspired Generations: His syntheses are considered classics, studied and admired for their brilliance. |
Export to Sheets
In essence:
Woodward’s mastery of total synthesis stemmed from his unique ability to combine profound theoretical understanding, strategic brilliance, innovative thinking, and (through his lab) exceptional experimental execution to build some of the most complex molecules known to humankind from simple starting materials. He didn’t just synthesize molecules; he designed the process with unparalleled elegance and foresight.
Woodward Strategic Planning: His syntheses were characterized by elegant and innovative strategies involving careful planning, novel reactions, and a deep understanding of reaction mechanisms. He approached synthesis like a complex chess game. Table.
Okay, based on the provided description, let’s break down the key elements of Robert Burns Woodward’s strategic planning in organic synthesis into a table.
| Element of Strategic Planning | Description / How Woodward Applied It |
| Overall Approach (“Chess Game”) | Viewed total synthesis as a complex intellectual puzzle, akin to a chess game. Required deep thinking, foresight, anticipation of potential problems, and evaluating multiple potential “moves” (reaction pathways) before committing to a course of action. |
| Rigorous Analysis & Planning | Meticulously planned synthetic routes before extensive laboratory work began. This involved a detailed analysis of the target molecule’s structure, identifying key bonds to form, and considering potential pitfalls and side reactions at each stage. |
| Elegance and Innovation | Sought pathways that were not merely functional but also elegant – characterized by efficiency, logic, and often novel or counter-intuitive chemical steps. He aimed for intellectual beauty in his synthetic designs, not just reaching the target molecule. |
| Use/Development of Novel Reactions | Did not limit himself to established reactions. He frequently employed newly developed reactions or modified existing ones creatively to achieve specific transformations that were crucial for his strategy, sometimes contributing to reaction methodology development itself. |
| Deep Mechanistic Understanding | His strategic planning was built upon a profound understanding of how chemical reactions work at a mechanistic level. This allowed him to predict reaction outcomes accurately, control stereochemistry, select appropriate reagents, and troubleshoot unexpected results effectively. |
| Control of Stereochemistry | Masterfully controlled the three-dimensional arrangement of atoms (stereochemistry) throughout complex sequences. This often required sophisticated planning to set multiple chiral centers correctly, a major challenge in complex molecule synthesis. |
| Strategic Bond Disconnections | Employed retrosynthetic analysis (though perhaps not always formally termed as such then), mentally breaking down the complex target molecule into simpler precursors to identify strategic bond disconnections and key intermediates. |
Export to Sheets
Key Takeaways:
- Intellectual Exercise: Woodward treated synthesis as a high-level intellectual exercise, not just a laboratory procedure.
- Foresight: His success relied heavily on his ability to anticipate problems and plan many steps.
- Creativity: He combined deep knowledge with creativity to devise innovative solutions to complex synthetic problems.
- Rigour: His approach was characterized by exceptional rigor in both planning and execution.
This table highlights the key components of Woodward’s renowned strategic approach to organic synthesis, emphasizing the planning, innovation, and deep understanding that made his work groundbreaking and influential. He didn’t just do synthesis; he designed it with exceptional skill and foresight.
Woodward Key Syntheses: Achieved the total synthesis of many complex natural products, including: <br> – Quinine (1944) – an anti-malarial drug <br> – Cholesterol and Cortisone (1951) – steroids <br> – Reserpine (1956) – a tranquilizer <br> – Chlorophyll (1960) – the pigment responsible for photosynthesis <br> – Vitamin B12 (1972) – a very complex molecule, a collaborative effort with Albert Eschenmoser. Table.
Okay, here is the information on Robert Burns Woodward’s key syntheses presented in a table format:
| Molecule Name | Year Synthesis Completed/Published | Significance / Type of Molecule |
| Quinine | 1944 | Anti-malarial drug; demonstrated the power of synthesis for creating complex, biologically active molecules. |
| Cholesterol | 1951 | An important steroid, a key molecule in biology, demonstrated mastery over complex fused ring systems. |
| Cortisone | 1951 | Medically important steroid (anti-inflammatory); demonstrated mastery over complex fused ring systems; achieved shortly after cholesterol. |
| Reserpine | 1956 | Complex alkaloid; used as an antipsychotic and antihypertensive drug (tranquilizer); showcased an elegant synthetic strategy. |
| Chlorophyll a | 1960 | The primary photosynthetic pigment in plants; the synthesis of its highly complex porphyrin structure was a major achievement. |
| Vitamin B12 | 1972 | It is an extremely complex vitamin and coenzyme, one of the most complex molecules ever synthesized, and a landmark collaborative effort with Albert Eschenmoser. |
Export to Sheets
Key Points about Woodward’s Syntheses:
- Complexity: These syntheses involved tackling molecules of increasing structural complexity.
- Strategy and Elegance: They were renowned for achieving the target and the elegance and strategic planning involved in the synthetic route.
- Impact: They proved that even the most complex natural products could be constructed in the laboratory, advanced the field of organic synthesis methodology, and sometimes had practical implications (e.g., confirming structures, providing access to materials).
- Confirmation of Structure: In many cases, synthesis ultimately confirmed a molecule’s proposed structure.
This table summarizes the major milestones in Woodward’s remarkable career in total synthesis.
Woodward-Hoffmann Rules (1965): Developed (with Roald Hoffmann) the Woodward-Hoffmann rules, which predict the stereochemistry (three-dimensional arrangement of atoms) of pericyclic reactions (a class of organic reactions). These rules are based on the symmetry properties of molecular orbitals. Table.
Okay, let’s put the details about the Woodward-Hoffmann Rules into a structured table format.
| Feature | Details |
| Rule Name | Woodward-Hoffmann Rules (or Rules of Orbital Symmetry) |
| Developers | Robert Burns Woodward and Roald Hoffmann |
| Year Developed | Formulated around 1965 (development and publication spanned a few years in the mid-1960s) |
| Purpose | To predict and explain the stereochemistry (the specific three-dimensional arrangement of atoms in the product) and the allowedness (whether a reaction proceeds easily under given conditions) of certain types of chemical reactions. |
| Class of Reactions | Pericyclic Reactions: Concerted reactions (bonds break and form simultaneously) that proceed through a cyclic transition state. Key examples include: <br> – Cycloadditions (e.g., Diels-Alder reaction) <br> – Electrocyclic reactions (ring opening/closing) <br> – Sigmatropic rearrangements |
| Theoretical Basis | Conservation of Orbital Symmetry: The rules are derived from the principle that the symmetry of the molecular orbitals involved in the reaction must be conserved throughout the reaction pathway. |
| Key Concepts | – Explains why certain pericyclic reactions proceed readily under thermal conditions (heat) while others require photochemical conditions (light) and vice versa. <br> – Predicts specific stereochemical outcomes (e.g., suprafacial vs. antarafacial addition, conrotatory vs. disrotatory ring opening/closing). |
| Impact/Significance | – Revolutionized the understanding of pericyclic reactions. <br> – Provided a powerful predictive tool for organic chemists planning syntheses. <br> – Unified experimental observations with fundamental quantum mechanical principles (orbital symmetry). <br> – Considered a landmark achievement in theoretical organic chemistry. <br> – Roald Hoffmann received the Nobel Prize in Chemistry in 1981 (shared with Kenichi Fukui for related work); Woodward had already received his Nobel in 1965 for synthesis but likely would have shared this one had he lived. |
Export to Sheets
This table summarizes the essential aspects of the Woodward-Hoffmann rules: who developed them, when, what they predict, the reactions they apply to, their theoretical foundation, and their profound impact on the field of organic chemistry.
Ahmed Zewail; Femtochemistry

Zewail in 2010
(Wiki Image By Science History Institute, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=33429523)
Ahmed Zewail Quotes. Table
Okay, let’s organize a selection of quotes from Ahmed Zewail, the “father of femtochemistry,” into a table format. These quotes reflect his pioneering work, his scientific philosophy, and his broader views on science and society.
| Quote Number | Quote | Source | Significance/Explanation |
| 1 | “Just as geographical exploration enriched humankind’s knowledge of the world, chemical exploration using femtosecond lasers is enriching our knowledge of the world of molecules.” | Nobel Lecture (1999) | Explains Femtochemistry’s Impact: Draws an analogy to exploration, highlighting how femtochemistry opened up a new frontier for observing the previously unseen molecular world during chemical reactions. |
| 2 | “With femtoscopy, we can see the dance of molecules in real time.” | Widely used description; attributed/representative | Captures the Essence of Femtochemistry: A simple, evocative way to describe the ability to observe the movement and interaction of atoms and molecules on the femtosecond timescale, revealing the dynamics of chemical change. |
| 3 | “The transition state is the heart of chemistry.” | Attributed/Representative | Highlights the Importance of Transition States: Emphasizes the significance of the fleeting, high-energy intermediate states that molecules pass through during a reaction, which femtochemistry has made directly observable. |
| 4 | “Curiosity is the key to creativity.” | Widely attributed | Reflects his Scientific Motivation: Underscores his belief that fundamental curiosity drives scientific discovery and innovation. |
| 5 | “Science and technology are the engines of progress.” | He expressed his views often. | Belief in Science for Societal Advancement: Highlights his strong belief in the power of science and technology to drive economic development, improve human well-being, and advance societies. |
| 6 | “The hope is that science will help to achieve peace and prosperity for all humankind.” | Reflects his broader humanitarian goals | Science for Humanity: He argues that science should serve a larger purpose, contributing to global peace and prosperity. This view aligns with his later role as a science envoy. |
| 7 | “We developed methods for studying chemical reactions on the time scale on which the reactions actually occur – femtoseconds.” | Explaining his work | Concise Summary of Achievement: A clear statement summarizing the core technical breakthrough of femtochemistry – achieving the necessary time resolution to observe reactions as they happen. |
| 8 | “My dream is to see Egypt become a modern, scientific society.” | Representative of his advocacy for Egypt | Commitment to his Homeland: This reflects his dedication to promoting science and education in Egypt and the broader Middle East. |
Export to Sheets
Key Takeaways from the Quotes:
- Observing the Unseen: Zewail emphasized the ability of femtochemistry to make the invisible dynamics of chemical reactions visible.
- Fundamental Curiosity: He highlighted curiosity as the driving force behind scientific discovery.
- Science for Progress: He believed strongly in the power of science and technology to advance human society.
- Bridging Cultures: His identity as both Egyptian and American informed his desire to use science as a bridge between cultures.
This table provides a structured overview of key statements reflecting Ahmed Zewail’s scientific contributions, motivations, and worldview. It highlights his pioneering role in femtochemistry and his belief in the power of science to transform our understanding of the world and improve human lives.
Ahmed Zewail YouTube Video
- Ahmed Zewail: Seeing with Electrons in Four Dimensions by MoleCluesTV: 65,862 views (http://www.youtube.com/watch?v=6KJxlf-HI3E)
- Ahmed Zewail: Year of Light can open doors to education by SPIETV: 46,100 views (http://www.youtube.com/watch?v=dmXcUiS71v8)
- 2014: Dr. Ahmed Zewail by Simon Fraser University: 37,442 views (http://www.youtube.com/watch?v=sCFYcF6B3A8)
- Ahmed Zewail – We Live on One Planet by NobelityProject: 14,342 views (http://www.youtube.com/watch?v=IabAClZw4bA)
- “The last 20 minutes of peace.” Ahmed Zewail on Being Awarded the 1999 Nobel Prize in Chemistry by Nobel Prize: 12,222 views (http://www.youtube.com/watch?v=kEPTOQujaP4)
pump-probe technique
Ahmed Zewail History

Zewail with his colleagues at the Zewail City of Science, Technology and Innovation in 2012
(Wiki Image By CFP-Zewail – Own work, CC BY-SA 4.0, https://commons.wikimedia.org/w/index.php?curid=61692419)
Ahmed Zewail (1946-2016) was an Egyptian-American chemist considered the “father of femtochemistry.” He pioneered ultrashort laser pulses to study chemical reactions in real time, allowing scientists to observe the breaking and forming of chemical bonds. His work revolutionized the field of chemical dynamics and earned him the Nobel Prize in Chemistry in 1999.
- Early Life and Education (1946-1969):
- Born in Damanhur, Egypt (1946): Raised in a middle-class family.
- Early Interest in Science: He showed an early interest in science and mathematics.
- University of Alexandria (B.Sc., M.Sc.): He earned his Bachelor of Science and Master of Science degrees in chemistry from Alexandria University in Egypt.
- Move to the United States (1969): He moved to the United States to pursue his doctoral studies.
- University of Pennsylvania (Ph.D., 1974): Earned his Ph.D. in chemistry from the University of Pennsylvania, working under the supervision of Robin M. Hochstrasser. His doctoral research focused on laser spectroscopy.
- Early Career and Development of Femtochemistry (1974-1990s):
- Postdoctoral Research at UC Berkeley (1974-1976): Worked as an IBM postdoctoral fellow at the University of California, Berkeley, with Charles B. Harris. This is where he began to explore using lasers to study ultrafast processes.
- California Institute of Technology (Caltech) (1976): He joined the faculty at Caltech, where he would spend the rest of his career.
- The Challenge of Observing Chemical Reactions: Before Zewail’s work, chemical reactions were studied primarily by analyzing the reactants and products. The actual process of bond breaking and formation, which occurs on incredibly fast timescales (femtoseconds – quadrillionths of a second), was considered impossible to observe directly.
- Pioneering Femtosecond Spectroscopy: Zewail pioneered the development of femtosecond spectroscopy, using ultrashort laser pulses to initiate and observe chemical reactions in real time.
- “Pump-Probe” Technique: He developed the “pump-probe” technique:
- A very short “pump” laser pulse initiates the reaction (breaks a chemical bond, for example).
- A second, even shorter “probe” laser pulse, delayed by a precisely controlled time interval (femtoseconds), interacts with the molecules and provides a “snapshot” of their state at that instant.
- By varying the delay between the pump and probe pulses, a “movie” of the reaction can be created, showing the movement of atoms and molecules during the reaction.
- Molecular Spectroscopy: Used molecular spectroscopy to analyze the signals from the probe pulse, providing information about the structure and energy of the molecules at different stages of the reaction.
- “Pump-Probe” Technique: He developed the “pump-probe” technique:
- First Observations of Transition States: Zewail and his team were the first to directly observe the transition states of chemical reactions directly. These fleeting, high-energy intermediates exist for only a few femtoseconds during the reaction process. This was a revolutionary breakthrough.
- Studies of Various Reactions: They applied femtosecond spectroscopy to study a wide range of chemical reactions, including:
- Bond breaking and formation
- Molecular vibrations and rotations
- Energy transfer processes
- Isomerization reactions
- Nobel Prize and Later Career (1999-2016):
- Nobel Prize in Chemistry (1999): Awarded the Nobel Prize in Chemistry “for his studies of the transition states of chemical reactions using femtosecond spectroscopy.” This recognized the fundamental importance of his work in revolutionizing chemical dynamics.
- Continued Research: Continued to develop and apply femtochemistry techniques to study increasingly complex systems, including biological molecules and materials.
- Director of the NSF Laboratory for Molecular Sciences (LMS):
- Director of the Physical Biology Center for Ultrafast Science and Technology
- Science Envoy: Served as a science envoy for the United States, promoting science and technology in the Middle East and worldwide.
- Advocate for Science Education: He was a strong advocate for science education and for supporting scientific research in developing countries.
- US Science Envoy
- Professor at Caltech.
- Death and Legacy:
- Death (2016): Died on August 2, 2016, at 70.
- “Father of Femtochemistry”: Widely regarded as the “father of femtochemistry,” a field he created and that continues to thrive.
- Revolutionized Chemical Dynamics: His work fundamentally changed how chemists study and understand chemical reactions. He provided a new way of seeing chemical processes, allowing scientists to observe the dynamics of bond breaking and formation.
- Impact on Multiple Fields: Femtochemistry has had a broad impact on many areas of chemistry, physics, biology, and materials science.
- Inspiration to Scientists: His groundbreaking work and his passion for science inspired generations of scientists.
- Technological Developments: Advanced lasers.
Ahmed Zewail’s development of femtochemistry was a major scientific breakthrough, opening up a new window into the ultrafast world of chemical reactions. He was a visionary scientist, a dedicated teacher, and a passionate advocate for science and education. His legacy continues to shape research in chemistry and related fields, and his work remains a testament to the power of human curiosity and ingenuity. He didn’t just make a discovery; he created a new field of science.
Ahmed Zewail, Femtochemistry, and Legacy. Table

Zewail in 1986
(Wiki Image By Unknown author – The Big T (yearbook of the California Institute of Technology), Public Domain, https://commons.wikimedia.org/w/index.php?curid=77882252)
Let’s detail Ahmed Zewail’s (1946-2016) work and legacy in a structured table format, focusing on his pioneering development of femtochemistry. This will cover the context, his key contributions, the technique itself, and the impact of his work.
| Aspect | Details | Significance |
| Context (Pre-Zewail) | – Chemical Reactions Understood Statistically: Before femtochemistry, chemical reactions were typically studied by measuring reaction rates and analyzing the reactants and products. The bond breaking and formation (occurring on incredibly fast timescales) was inaccessible. <br> – Transition States: Chemists understood that reactions proceed through transition states (high-energy, short-lived intermediates), but these could only be inferred, not directly observed. | – There was a fundamental gap in understanding the real-time dynamics of chemical reactions at the atomic and molecular level. |
| Zewail’s Innovation: Femtochemistry | – Ultrashort Laser Pulses: Zewail pioneered using ultrashort laser pulses (femtoseconds – 10⁻¹⁵ seconds, or one quadrillionth of a second) to initiate and observe chemical reactions in real-time. <br> – “Pump-Probe” Technique: Developed the “pump-probe” technique: <br> – A “pump” pulse initiates the reaction. <br> – A “probe” pulse, delayed by a precisely controlled time interval, interacts with the molecules and provides a “snapshot” of their state at that instant. <br> – By varying the delay between the pump and probe pulses, a “movie” of the reaction can be created. <br> – Molecular Spectroscopy: Used molecular spectroscopy to analyze the signals from the probe pulse, providing information about the structure and energy of the molecules at different stages of the reaction. | – Revolutionized the study of chemical dynamics. This allowed scientists to observe the breaking and forming of chemical bonds, previously thought impossible. <br> – Opened up a new field of chemistry: femtochemistry. |
| Key Discoveries and Applications | – Observation of Transition States: Observed the transition states of chemical reactions, providing direct evidence for their existence and allowing for detailed study of their properties. <br> – Real-Time Dynamics: Studied the real-time dynamics of a wide range of chemical reactions, including: <br> – Bond breaking and formation <br> – Molecular vibrations and rotations <br> – Energy transfer processes <br> – Isomerization reactions <br> – Applications in: <br> – Understanding fundamental chemical processes <br> – Controlling chemical reactions with light <br> – Materials science <br> – Biology (e.g., studying photosynthesis, DNA dynamics) | – Provided unprecedented insights into the mechanisms of chemical reactions. <br> – Opened up new possibilities for controlling chemical reactions with light. <br> – Broadly impacted many chemistry, physics, and biology areas. |
| Recognition and Awards | – Nobel Prize in Chemistry (1999): Awarded “for his studies of the transition states of chemical reactions using femtosecond spectroscopy.” <br> – Numerous other awards and honors. | – International recognition of the groundbreaking nature of his work. <br> – solidified his place as a pioneer in chemistry. |
| Legacy | – “Father of Femtochemistry”: Considered the founder of femtochemistry, a vibrant and rapidly growing field. <br> – New Way of Seeing: Provided a fundamentally new way to observe and understand chemical reactions. <br> – Inspiration to Scientists: Inspired a generation of scientists to pursue research in ultrafast phenomena. <br> – Continued Development: Femtochemistry techniques continue to be refined and applied to an increasingly wide range of scientific problems. <br> – Technological Advancements: His work spurred the development of faster and more sophisticated laser technology. | – His work transformed our understanding of chemical dynamics. <br> – Femtochemistry is now an established and essential tool for studying chemical reactions at the most fundamental level. <br> – His legacy continues to shape research in chemistry and related fields. |
Export to Sheets
Key Takeaways:
- Ultrafast Timescale: Zewail’s work opened the femtosecond timescale for chemical investigation, allowing scientists to observe the incredibly fast bond-breaking and formation processes.
- “Pump-Probe” Technique: His “pump-probe” technique is the foundation of femtochemistry.
- Revolutionary Impact: Femtochemistry revolutionized the study of chemical dynamics, providing unprecedented insights into reaction mechanisms.
- Broad Applications: The techniques and concepts of femtochemistry have applications in many areas of science and technology.
This table comprehensively overviews Ahmed Zewail’s pioneering work in femtochemistry, highlighting the context, key innovations, applications, and lasting legacy. It emphasizes the revolutionary nature of his contribution and its impact on our understanding of chemical reactions. The table format allows for a clear and structured presentation of this complex and vital scientific advancement.
Zewail Ultrashort Laser Pulses: Zewail pioneered using ultrashort laser pulses (femtoseconds—10⁻¹⁵ seconds, or one quadrillionth of a second) to initiate and observe chemical reactions in real time. Table.
You’re right to emphasize the groundbreaking nature of Ahmed H. Zewail’s work with ultrashort laser pulses. Here’s a table summarizing his contributions:
Ahmed H. Zewail: Ultrashort Laser Pulses and Femtochemistry
| Aspect | Description | Significance |
| Femtosecond Laser Technology | * Developed techniques to generate and utilize ultrashort laser pulses with durations in the femtosecond (10⁻¹⁵ seconds) range. <br> * This enabled the initiation and observation of chemical reactions on the timescale of atomic motion. | * Provided the necessary tool to “freeze” molecular motion and observe chemical reactions as they happen. <br> * Revolutionized the study of chemical reaction dynamics. <br> * Created the field of femtochemistry. |
| Real-Time Observation of Reactions | * Enabled the direct observation of transition states and intermediate structures during chemical reactions. <br> * Allowed researchers to “see” the breaking and forming of chemical bonds in real time. | * Provided unprecedented insights into chemical reaction mechanisms. <br> * Allowed for the detailed study of how molecules change during chemical transformations. <br> * Increased the fundamental understanding of chemical reaction dynamics. |
| Femtochemistry | * Established the field of femtochemistry, the study of chemical reactions on the femtosecond timescale. <br> * Pioneered the use of pump-probe spectroscopy to observe molecular dynamics. | * Opened up a new era of chemical research. <br> * Transformed our understanding of chemical reactivity. <br> * Has applications in many different fields, including biochemistry. |
| Impacts on Numerous Fields | Femtochemistry techniques have increased the understanding of various fields, including materials science, photochemistry, and biochemistry. | Numerous scientific fields have greatly benefitted by providing such clear data on chemical reactions. |
| Nobel Prize | Awarded the 1999 Nobel prize in chemistry. | Recognized the importance of this work and how it has furthered the field of chemistry. |
Export to Sheets
Key Points:
- “High-Speed Molecular Photography”: Zewail’s work provided the means to take “snapshots” of molecules during reactions.
- Unprecedented Detail: His techniques allowed chemists to study reaction dynamics with unprecedented temporal resolution.
- Fundamental Discoveries: Femtochemistry has led to deep insights into the mechanisms of chemical reactions.
- Broad Applications: The techniques are used in a variety of scientific disciplines.
Zewail “Pump-Probe” Technique: Developed the “pump-probe” technique: <br> – A “pump” pulse initiates the reaction. <br> – A “probe” pulse, delayed by a precisely controlled time interval, interacts with the molecules and provides a “snapshot” of their state at that instant. <br> – By varying the delay between the pump and probe pulses, a “movie” of the reaction can be created. Table.
You’ve accurately described the “pump-probe” technique developed by Ahmed H. Zewail. It’s a key component of femtochemistry. Here’s a table summarizing the technique:
Ahmed H. Zewail: “Pump-Probe” Technique
| Aspect | Description | Significance |
| Pump Pulse | * A short laser pulse (“pump”) initiates the chemical reaction by exciting the reactant molecules. <br> * This provides the initial energy needed to drive the reaction. | * Allows the controlled initiation of chemical reactions on a femtosecond timescale. <br> * Creates a synchronized starting point for observing the reaction dynamics. <br> * Start time = zero. |
| Probe Pulse | * A second, delayed laser pulse (“probe”) interacts with the molecules at a specific time after the pump pulse. <br> * The probe pulse measures a property of the molecules, such as their absorption or emission of light. | * Provides a “snapshot” of the molecular state at a specific point in time. <br> * The delay of the probe pulse determines the “frame” of the molecular movie. <br> * Allows scientists to directly view the chemical process at specific time intervals. |
| Varying Delay | * By systematically varying the time delay between the pump and probe pulses, a series of snapshots are obtained. <br> * These snapshots are then compiled to create a “movie” of the chemical reaction. | * Enables the construction of a dynamic picture of the reaction pathway. <br> * Allows the observation of transition states and intermediates that are otherwise impossible to detect. <br> * By compiling many snapshots, a movie can be created, showing the entire chemical reaction as it happens. |
| “Molecular Movie” | By taking many “snapshots” of the chemical reaction and compiling them into a single file, a “molecular movie” is created. | “Molecular movies” give insight into how chemical bonds are formed and broken. |
Export to Sheets
Key Points:
- Time-Resolved Measurements: The technique provides time-resolved measurements of chemical reactions.
- Visualizing Molecular Motion: It enables the creation of “molecular movies” that illustrate the dynamics of chemical processes.
- Understanding Reaction Pathways: It allows for the detailed study of reaction pathways and intermediate structures.
- Fundamental Technique: This “pump-probe” technique is still used in modern research.
Zewail Molecular Spectroscopy: Used molecular spectroscopy to analyze the signals from the probe pulse, providing information about the structure and energy of the molecules at different stages of the reaction. Table.
You’ve accurately pointed out the crucial role of molecular spectroscopy in Ahmed H. Zewail’s femtochemistry. Here’s a table detailing how he used this technique:
Ahmed H. Zewail: Molecular Spectroscopy in Femtochemistry
| Aspect | Description | Significance |
| Probe Pulse Interaction | * The probe laser pulse interacts with the molecules at a specific time after the pump pulse. <br> * This interaction induces changes in the molecules’ energy levels. | * Allows the interrogation of the molecular state at a precise moment during the reaction. <br> * Provides information about the molecules’ structure and energy distribution. <br> * The interaction between the probe pulse and the molecule is recorded. |
| Spectroscopic Analysis | * The signals from the probe pulse are analyzed using molecular spectroscopy techniques (e.g., absorption spectroscopy, emission spectroscopy, Raman spectroscopy). <br> * This analysis reveals information about the molecules’ vibrational and electronic states. | * Provides a detailed picture of the molecules’ structure and energy levels at different stages of the reaction. <br> * Enables the identification of transient species and the determination of their properties. <br> * Allows scientists to see the changes in molecular configuration as the reaction proceeds. |
| Structural and Energetic Information | * By analyzing the spectroscopic signals, researchers can determine the geometry of the molecules, the lengths and angles of chemical bonds, and the distribution of energy among the molecules’ vibrational modes. | * Provides a comprehensive understanding of the molecular dynamics during the reaction. <br> * Allows the construction of a detailed “molecular movie” showing the structural and energetic changes over time. <br> * This information allows for a much greater understanding of the reaction mechanism. |
| Time Resolved Data | The spectroscopic data is time resolved, meaning it is collected at specific time intervals. This allows for the data to be placed in the correct time line. | The time-resolved data is what allows the creation of “molecular movies” of chemical reactions. |
Export to Sheets
Key Points:
- Detailed Molecular Information: Molecular spectroscopy provides detailed information about the structure and energy of molecules.
- Transient Species: It enables the identification and characterization of short-lived intermediate species.
- Reaction Dynamics: It allows for the study of how molecular structure and energy change during a reaction.
- Precision Measurements: The combination of ultrashort laser pulses and molecular spectroscopy allows for very precise measurements of molecular dynamics.
Zewail Observation of Transition States: I observed the transition states of chemical reactions, providing direct evidence for their existence and allowing for a detailed study of their properties. Table.
You’ve summarized Ahmed H. Zewail’s contribution very accurately. His ability to observe transition states was a monumental leap in understanding chemical reactions. Here’s a table that emphasizes this aspect of his work:
Ahmed H. Zewail: Observation of Transition States
| Aspect | Description | Significance |
| Direct Observation | * Using femtosecond laser spectroscopy, Zewail was able to directly observe the transition states of chemical reactions. <br> * These transient structures, which are fleeting intermediates between reactants and products, had previously only been theorized. | * Provided concrete experimental evidence for the existence of transition states. <br> * Confirmed theoretical models of reaction pathways. <br> * Enabled the direct study of these ephemeral structures. |
| Characterization of Properties | * Zewail’s techniques allowed for the determination of the properties of transition states, such as their geometry, energy levels, and lifetimes. <br> * These properties are crucial for understanding reaction mechanisms. | * Provided crucial details about reaction dynamics. <br> * Enhanced understanding of how chemical bonds are formed and broken. <br> * Allowed for very detailed chemical modelling to be made. |
| Detailed Reaction Mechanisms | * By mapping the evolution of transition states, Zewail elucidated the detailed mechanisms of chemical reactions. <br> * Revealed the precise sequence of atomic motions during the reaction. | * Transformed our understanding of chemical reactivity. <br> * Provided a deeper insight into how molecules transform during reactions. <br> * Revolutionized the field of reaction kinetics. |
| Impact on reaction dynamics. | By observing these transition states, scientist where able to gain data that was previously unobtainable, and therefore where able to create more accurate chemical models. | Increased our understanding of chemical reactions. |
Export to Sheets
Key Points:
- Visualizing the Unseen: Zewail’s work made it possible to “see” what was previously only imagined.
- Confirming Theory: His experimental observations validated existing theoretical models.
- Unlocking Reaction Secrets: It helped reveal the intricate dance of atoms during chemical transformations.
- Detailed modeling: Increased the ability to perform accurate chemical modeling.
Zewail Real-Time Dynamics: Studied the real-time dynamics of a wide range of chemical reactions, including: <br> – Bond breaking and formation <br> – Molecular vibrations and rotations <br> – Energy transfer processes <br> – Isomerization reactions. Table.
You’ve accurately outlined the broad scope of Ahmed H. Zewail’s research. His work allowed for the real-time observation of numerous fundamental chemical processes. Here’s a table summarizing these areas:
Ahmed H. Zewail: Real-Time Dynamics of Chemical Reactions
| Reaction Type | Description | Significance |
| Bond Breaking and Formation | * Observed the precise moments when chemical bonds break and form during reactions. <br> * Mapped the pathways of these bond rearrangements. | * Provided direct evidence for the mechanisms of chemical transformations. <br> * Revealed the energy barriers and transition states involved in these processes. <br> * Allowed a clear view of molecular change during a reaction. |
| Molecular Vibrations and Rotations | * Studied the real-time motions of atoms within molecules, including their vibrations and rotations. <br> * Measured the rates of these motions. | * Provided insight into the fundamental dynamics of molecular behavior. <br> * Helped to understand how energy is distributed within molecules. <br> * Allowed observation of energy transfer in molecules. |
| Energy Transfer Processes | * Observed how energy flows between different parts of molecules during reactions. <br> * Measured the rates of energy transfer between vibrational and electronic states. | * Explained how energy is channeled within molecules to drive chemical reactions. <br> * Important for understanding processes like photosynthesis and combustion. <br> * Showed energy flow on the molecular level. |
| Isomerization Reactions | * Studied the dynamics of isomerization reactions, where molecules rearrange their atoms into different configurations. <br> * Observed the pathways and timescales of these rearrangements. | * Provided a detailed understanding of how molecules change their shapes. <br> * Important for understanding biological processes and chemical synthesis. <br> * Allowed an observation of molecular shape change in real time. |
| Solvation Dynamics | Zewail also contributed significantly to understanding how solvent molecules interact with solute molecules during chemical reactions. | This helped to uncover how solvents affect reaction dynamics. |
Export to Sheets
Key Points:
- Comprehensive Study: Zewail’s work encompassed a wide range of fundamental chemical processes.
- Time-Resolved Insights: He provided time-resolved data that revealed the dynamics of these processes.
- Fundamental Mechanisms: His research elucidated the fundamental mechanisms of chemical reactions.
- Wide Applicability: The findings have broad implications across numerous fields of chemistry.
Zewail Applications in: Understanding fundamental chemical processes <br> – Controlling chemical reactions with light <br> – Materials science <br> – Biology (e.g., studying photosynthesis, DNA dynamics). Table.
Ahmed H. Zewail’s work with femtochemistry has had a broad impact, extending beyond fundamental chemistry to various applied fields. Here’s a table summarizing some of these applications:
Ahmed H. Zewail: Applications of Femtochemistry
| Application Area | Description | Significance |
| Fundamental Chemical Processes | * Provides detailed insights into reaction mechanisms, transition states, and molecular dynamics. <br> * Elucidates the role of energy transfer, bond breaking/formation, and molecular rearrangements. | * Deepens our basic understanding of chemical reactions at the molecular level. <br> * Refines chemical theories and models. <br> * Essential for developing new chemical synthesis techniques. |
| Controlling Chemical Reactions with Light | * Explores the use of ultrashort laser pulses to selectively control chemical reactions. <br> * Investigates how light can be used to manipulate molecular pathways. | * Opens possibilities for controlling chemical reactions with greater precision. <br> * Potential applications in fields like photochemistry, catalysis, and materials synthesis. <br> * Allows control of chemical reactions that were previously impossible. |
| Materials Science | * Study the dynamics of processes in materials, such as phase transitions, surface reactions, and energy transport. <br> * Helps understand the properties and behavior of materials at the nanoscale. | * Enables the development of new materials with tailored properties. <br> * Provides insights into the fundamental processes that govern material behavior. <br> * Allows for the creation of new techniques in nanotechnology. |
| Biology | * Investigates biological processes at the molecular level, such as photosynthesis, protein folding, DNA dynamics, and enzyme catalysis. <br> * Reveals the mechanisms of biological reactions in real time. | * Provides deeper insights into fundamental life processes. <br> * Opens possibilities for designing new drugs and understanding disease mechanisms. <br> * Helps increase our understanding of DNA replication, protein folding, and photosynthesis. |
Export to Sheets
Key Points:
- Interdisciplinary Impact: Femtochemistry has applications across various scientific disciplines.
- Molecular-Level Control: The technology allows for precise manipulation of chemical reactions.
- Enhanced Understanding: It deepens our understanding of fundamental processes in chemistry and biology.
- Nanoscale Applications: It has become vital in many nanotechnology research projects.
Similarities to Gilbert N. Lewis, Linus Pauling, Dorothy Crowfoot Hodgkin, Robert Burns Woodward, and Ahmed Zewail
Okay, let’s create a table summarizing the key similarities shared by Gilbert N. Lewis, Linus Pauling, Dorothy Crowfoot Hodgkin, Robert Burns Woodward, and Ahmed Zewail.
| Aspect of Similarity | Explanation / How it Applies to the Group |
| 1. Fundamental Impact on Chemistry | All five revolutionary contributions fundamentally changed our understanding of chemistry in their respective fields (bonding, structure, synthesis, and dynamics). They didn’t just refine knowledge; they reshaped it. |
| 2. Focus on Molecular/Atomic Level | Their work universally centered on understanding matter at the molecular and atomic levels—how atoms bond, the precise three-dimensional structure of molecules, and how these structures change during chemical processes. |
| 3. Pioneering Techniques & Methodologies | They either developed or were masters at applying cutting-edge experimental or theoretical techniques for their time (e.g., Lewis structures, quantum mechanics, X-ray crystallography, complex synthesis, and femtochemistry). |
| 4. Tackling Complexity | They did not shy away from incredibly complex problems, whether the quantum nature of bonding, the intricate structures of large biomolecules, multi-step synthesis, or the fleeting moments of a chemical reaction. |
| 5. Interdisciplinary Impact | Their discoveries often transcended the traditional boundaries of chemistry, significantly impacting related fields like physics, biology, and medicine. |
| 6. High Level of Recognition | All have achieved significant recognition within the scientific community. Pauling, Hodgkin, Woodward, and Zewail received Nobel Prizes in Chemistry. Lewis, despite numerous nominations, is universally regarded as profoundly influential. |
| 7. Influence on Future Generations | Most were influential educators and mentors who trained subsequent generations of leading scientists, extending their impact through the people they taught and inspired. |
Export to Sheets
In Summary:
Despite specializing in different areas (bonding theory, quantum chemistry, structural biology, organic synthesis, femtochemistry), these five chemists share common threads of being revolutionary thinkers, focusing on the fundamental molecular level, pioneering new methods, tackling immense complexity, having interdisciplinary influence, achieving high recognition, and shaping the future of chemistry through their work and mentorship. They represent the pinnacle of 20th-century chemical innovation.
Difference Gilbert N. Lewis, Linus Pauling, Dorothy Crowfoot Hodgkin, Robert Burns Woodward, and Ahmed Zewail
Okay, let’s create a comparison table highlighting the key differences between Gilbert N. Lewis, Linus Pauling, Dorothy Crowfoot Hodgkin, Robert Burns Woodward, and Ahmed Zewail. This focuses on their distinct areas of expertise, methodologies, and the nature of their major contributions.
| Feature | Gilbert N. Lewis (1875-1946) | Linus Pauling (1901-1994) | Dorothy Crowfoot Hodgkin (1910-1994) | Robert Burns Woodward (1917-1979) | Ahmed Zewail (1946-2016) |
| Primary Field/Specialty | Physical Chemistry (Bonding, Thermodynamics, Acids/Bases) | Physical/Quantum Chemistry, Structural Biology | Structural Chemistry/Biochemistry (X-ray Crystallography) | Organic Chemistry (Synthesis) | Physical Chemistry (Chemical Dynamics, Femtochemistry) |
| Key Contribution | Concept of Covalent Bond / Lewis Structures / Lewis Acid-Base Theory | Nature of the Chemical Bond (using Quantum Mechanics) / Protein Structures (alpha-helix, beta-sheet) | Structure Determination of Complex Biomolecules (Penicillin, Vitamin B12, Insulin) | Total Synthesis of Complex Natural Products / Woodward-Hoffmann Rules | Development of Femtochemistry / Observation of Transition States in Real-Time |
| Primary Methodology | Conceptual/Theoretical (based on data) | Mix of Theory (Quantum Mechanics) and Experiment (X-ray Diffraction) | Primarily Experimental (Master of X-ray Crystallography) | Primarily Experimental (Master of Organic Synthesis), also Theoretical (W-H Rules) | Primarily Experimental (Development/Use of Ultrafast Laser Spectroscopy) |
| Main Focus | Static electron arrangement and bonding | Static structure and bonding (molecules & biomolecules) | Static 3D structure of complex molecules | Building complex static structures; understanding reaction pathways | Dynamic processes: observing reactions and transition states in time |
| Type of Molecules Studied | General chemical bonding concepts | Minerals, simple molecules, proteins, DNA (attempted) | Large, complex biomolecules (Penicillin, B12, Insulin) | Complex organic natural products | Primarily small molecules during reactions, expanding later |
| Era of Major Breakthroughs | 1910s-1920s | 1930s-1950s | 1940s-1960s | 1940s-1970s | 1980s-1990s |
| Nobel Prize Status | Nominated 41 times, Never Won | Won (Chemistry, 1954; Peace, 1962) | Won (Chemistry, 1964) | Won (Chemistry, 1965) | Won (Chemistry, 1999) |
Export to Sheets
Key Distinctions Summarized:
- Lewis: Focused on the fundamental concept and representation of electron sharing (covalent bonds) and a broader definition of acids/bases. Primarily conceptual/theoretical.
- Pauling: Applied quantum mechanics to explain bonding and determined the fundamental structures of proteins. Bridged multiple fields.
- Hodgkin: Was the master experimentalist using X-ray crystallography to determine the exact 3D structures of incredibly complex biological molecules.
- Woodward: He was the master synthesizer, demonstrating the art and science of building complex organic molecules step-by-step, and contributed theory (Woodward-Hoffmann rules).
- Zewail: Focused on the dynamics of chemical reactions, developing techniques to observe bond breaking and formation on the femtosecond timescale – watching molecules move during reactions.
This table highlights the unique niche and specific groundbreaking contributions of each of these five highly influential chemists, showcasing the diversity of expertise within the field during the 20th century. While all focused on the molecular world, they asked different questions and developed various tools to answer them.
Compared to Gilbert N. Lewis, Linus Pauling, Dorothy Crowfoot Hodgkin, Robert Burns Woodward, and Ahmed Zewail
Okay, let’s compare these five giants of 20th-century chemistry, highlighting their distinct contributions and approaches:
- Gilbert N. Lewis stands as the conceptual architect of covalent bonding. He provided the foundational, intuitive picture of atoms sharing electron pairs (covalent bonds) and achieving stability (octet rule), brilliantly visualized through his Lewis structures. His work gave chemists a practical, non-mathematical way to understand how most molecules are held together, and his broad definition of acids and bases remains crucial. He focused on the fundamental rules of electron arrangement in static structures.
- Linus Pauling took Lewis’s ideas and provided a deeper quantum mechanical explanation. He applied the complex principles of quantum mechanics to chemistry, developing valence bond theory, hybridization, and resonance to explain the nature and geometry of chemical bonds with unprecedented rigor. Pauling uniquely bridged theoretical chemistry, experimental structure determination (using X-ray diffraction, like Hodgkin), and biology, famously elucidating the alpha-helix and beta-sheet structures of proteins. He was a master synthesizer of ideas across disciplines.
- Dorothy Crowfoot Hodgkin was the master experimentalist of structure determination. Using the demanding technique of X-ray crystallography, she tackled incredibly complex biological molecules that many thought were impossible to decipher. Her determination of the structures of penicillin, vitamin B12, and finally, insulin required decades of meticulous work and pushed the boundaries of crystallographic methods. She revealed the precise 3D architecture of life’s crucial molecules.
- Robert Burns Woodward was the master artist and strategist of organic synthesis. While Hodgkin revealed nature’s structures, Woodward built them. He demonstrated that even the most complex natural products (quinine, chlorophyll, vitamin B12) could be constructed step-by-step in the laboratory through elegant and innovative reaction pathways. His work transformed synthesis from a trial-and-error craft into a predictable science and art form, and he also contributed significantly to reaction theory with the Woodward-Hoffmann rules.
- Ahmed Zewail shifted the focus from static structure to chemical dynamics. He pioneered femtochemistry, developing ultrafast laser techniques to observe the fleeting transition states of chemical reactions – the actual process of bonds breaking and forming – on the femtosecond timescale. While the others primarily studied what molecules are, Zewail provided the tools to watch what molecules do in real time during their transformation.
In Comparison:
- Lewis vs. Pauling: Lewis provided the foundational concept of electron sharing; Pauling provided the quantum mechanical explanation and applied it bro
- Pauling vs. Hodgkin: Both used X-ray crystallography, but Pauling used it more broadly alongside theory to understand general principles of structure (like protein secondary structures), while Hodgkin focused intensely on solving the complete, specific structures of exceptionally complex individual molecules.
- Hodgkin vs. Woodward: Hodgkin determined the structure of complex natural molecules; Woodward synthesized them. They were masters of analysis and synthesis, respectively, often working on molecules of similar complexity (like B12).
- Zewail vs. Others: Zewail stands apart by focusing on the ultrafast dynamics of reactions, whereas the others were primarily concerned with static bonding and structure (though Woodward studied reaction pathways).
Together, these five scientists represent a massive leap in chemical understanding during the 20th century, covering the fundamental nature of bonds (Lewis, Pauling), the intricate architecture of life’s molecules (Pauling, Hodgkin), the power to construct complex organic matter (Woodward), and the ability to witness the very act of chemical change (Zewail).



